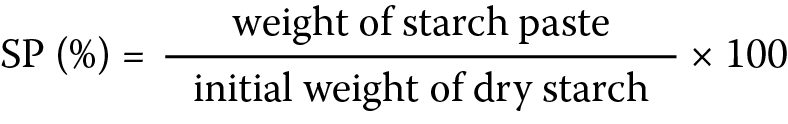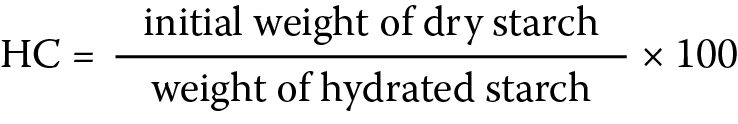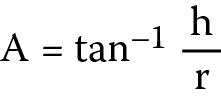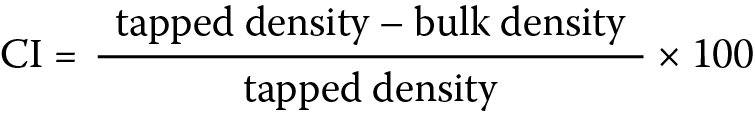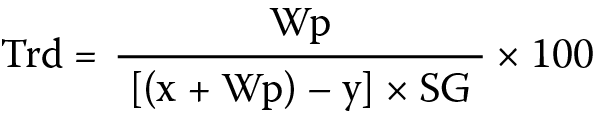Abstract
Background. Pre-gelatinization is one of the most common physical methods of starch modification, which involves heating to bring about significant changes in the nature of starch, such as high swelling, loss of crystallinity, solubility in cold water, and improved pasting.
Objectives. To evaluate the structural and physicochemical properties of starch from Neorautanenia mitis tubers, and determine the effect of pre-gelatinization on the functional properties of this starch.
Materials and methods. Properties of the pre-gelatinized starch (NMPS), such as flow, swelling power, hydration capacity, pH, morphology, Fourier-infrared spectroscopy (FTIR), differential scanning calorimetry, and pasting characteristics, were compared with those of the native starch (NMNS).
Results. Pre-gelatinized starch had good flow with the angle of repose at 33.69°. Carr’s index was 10.90% and 7.50%, and the Hausner ratio was 1.12 and 1.05 for NMNS and NMPS, respectively. Both starches had neutral to near-neutral pH (7.00 and 6.04, respectively). The hydration capacity of NMPS (59.00%) was about 2 times higher than that of NMNS (25.80%), while the swelling power of NMPS between 40°C and 60°C was higher than that of NMNS, and maximum swelling for both starches was observed at 80°C. Morphology showed that NMNS granules were discrete, smooth and spherical, while those of NMPS were aggregated, with rough surfaces. The FTIR spectra of both starches showed identical absorption peaks but the enthalpy of gelatinization differed for both starches. The pasting properties also varied significantly among the starch samples. Native starch had better peak viscosity, breakdown viscosity and pasting temperature, while NMPS presented better trough viscosity, final viscosity, setback viscosity, and pasting time.
Conclusions. The results showed that pre-gelatinized starch from N. mitis tubers possesses high swelling and hydration abilities and significant pasting properties, and may be used as a disintegrant in solid dosage formulations and in products requiring low viscosities and bond strength.
Key words: physicochemical properties, starch, structural, Neorautanenia mitis, pre-gelatinization
Background
Starch is, after cellulose, the most abundant natural carbohydrate found in plants. It can be found in different plant parts, including the seeds, roots, stems, leaves, and fruits, as a ready source of energy. Being commonly found in plants, it is relatively abundant, cheap, safe, degradable, and easily modifiable. As such, starch is a material widely used in food, cosmetics and pharmaceutical industries. However, starch in its native form has limited application in industrial processes because of some of its undesirable inherent properties, such as poor processability, poor solubility in common organic solvents, retrogradation, low shear stress resistance, and susceptibility to thermal decomposition.1 Modification processes, such as enzymatic hydrolysis, chemical modifications and physical modifications, have been employed to develop new functional properties and to improve the inherent properties of starch.2
Physical methods of starch modification, like heat gelatinization, are commonly utilized because they do not require the use of chemicals and thus, are deemed safe for human consumption.3 Pre-gelatinization is one of the most popular physical methods of starch modification and it involves heating a starch suspension at a temperature below its gelatinization temperature.4 The process of pre-gelatinization causes an irreversible disruption of the starch granules by reorganizing the hydrogen bonding within them. This process is responsible for several changes in starch properties including high swelling, loss of crystallinity, solubility in cold water, and improved pasting and flow.5 These properties make pre-gelatinized starch suitable for many processes in the pharmaceutical and food industries.6
Starches from various plant sources have their own unique properties that are utilized to meet specific needs. Starch from Neorautanenia mitis is examined in this study. Neorautanenia mitis (A. Rich) Verdcourt of the Fabaceae family is a leguminous subshrubby plant with a tuberous rootstock. It is usually found growing in grasslands, bushy lands and open woodlands and rocky soils of the central, southern and western regions of Africa.7 Traditionally, this plant was used as a fish poison and an insecticide, and to treat skin infections, syphilis and psychiatric conditions. Experimental studies have also shown that it possesses acaricidal, antimicrobial and antinociceptive properties.8, 9
Despite the therapeutic potentials of this plant, the starch that abounds in its tubers has not been documented to have nutritional value, nor has it been exploited for use as an excipient in the food, cosmetics or pharmaceutical industries. However, Olayemi et al. has examined the potential of starch from the tuberous roots of N. mitis to be used in tablet formulations.10
Objectives
In view of the fact that the traditional sources of starch are overexploited, tubers of N. mitis could be used as a new source of starch and starch derivatives. Therefore, the aim of this study is to prepare pre-gelatinized starch from tubers of N. mitis, and evaluate the structural, physicochemical and pasting properties of the modified starch, with a view to provide new starch sources with potential functional properties.
Materials and methods
The materials used included N. mitis tubers purchased from Suleja Community Market, Suleja, Nigeria. The N. mitis starch was prepared in the NIPRD Laboratory (Abuja, Nigeria). All other reagents used were of analytical grade.
Extraction of starch
Starch was extracted using a previously described method.10 Tubers of N. mitis were peeled, washed in water and diced. Then, they were soaked in a solution of sodium metabisulphite solution (0.75% w/v) for 2 h and ground using a blender. The blended mixture was sieved using a muslin cloth and the suspension was centrifuged at 1500 rpm for 15 min in a centrifuge (Heraeus Sepatech Labofuge Ae, Münster, Germany). The supernatant was discarded and the sediment (starch) was air-dried at room temperature for 24 h, and then pulverized in a mortar, packaged appropriately and stored in a desiccator.
Pre-gelatinization
of N. mitis native starch (NMNS)
A previously described method for pre-gelatinization was adopted.11 A starch slurry (20% w/v) was prepared in water and heated at 55°C in a water bath (Karl Kobb, Dreieich, Germany) with constant stirring for 15 min. The resulting paste was dried in an oven (Biobase Biodustry Co. Ltd., Shandong, China) at 60°C for 48 h and sieved using a 250-μm sieve mesh. The pre-gelatinized starch (NMPS) was stored in an airtight container and placed in a desiccator until further use.
Evaluation of NMNS and NMPS
Morphology
Starch samples (NMNS and NMPS) were mounted on metal stubs, coated with gold and analyzed using a scanning electron microscope (SEM; ASPEX 3020, PSEM 2; Field Electron and Ion, FEI Corp, Hilsboro, USA). Images of the starch surfaces were obtained at ×1000 magnification at a current of 7 mA for 90 s.
Fourier-transform infrared (FTIR) spectra studies
The starches (NMNS and NMPS) were triturated with potassium bromide powder and were made into pellets (1 ton/cm2). Infrared (IR) spectra were obtained between scanning ranges of 4000 cm−1 and 400 cm−1 from a Magna-IR 560 spectrometer (Perkin Elmer, Waltham, USA).
Gelatinization temperature
The gelatinization temperature was determined using a differential scanning calorimeter (DSC; Model DSC 204 F1; Netzsch, Selb, Germany). Starch samples were placed in the aluminum pans of the equipment and scanned at between 60°C and 300°C at a heating rate of 10°C/min under constant nitrogen flow.
pH determination
Slurries (5% w/v) of the starch samples were prepared in distilled water and the pH was measured at room temperature (28°C) using a pH meter (Metler Toledo, Greifensee, Switzerland). Triplicate determinations were made and the average value was recorded.
Swelling power
Starch slurries (1% w/v) were prepared with distilled water and heated in a water bath at 40°C for 30 min. The dispersions were centrifuged at 1500 rpm for 30 min, the supernatant was discarded, the weight of the starch paste was determined, and the swelling power (SP) was computed using the equation below (Equation 1):
This procedure was repeated at 50°C, 60°C, 70°C, 80°C, and 90°C for both starch samples.
Hydration capacity
The method of Kornblum and Stoopak12 was adopted to determine the starch hydration capacity (HC). Starch dispersions (1% w/v) were prepared with distilled water, poured into pre-weighed, stoppered centrifuge tubes and shaken intermittently for 10 min. The tubes were allowed to stand for another 10 min at room temperature and then centrifuged at 1500 rpm for 5 min. The supernatant was discarded, and the weight of the sediment (hydrated starch) was determined and used to compute the HC as shown below (Equation 2):
Flow properties
Angle of repose
The angle of repose was determined using the funnel method. Twenty grams of starch sample were allowed to flow through the orifice of a funnel clamped at a fixed height from a flat surface. The height (h) and radius (r) of the heap formed were measured and used to compute the angle of repose (A) as shown below (Equation 3):
Bulk and tapped densities
The volume occupied by the starch samples (50 g) in a graduated measuring cylinder was noted as the bulk volume and used to compute the bulk density [g/mL]. In the same way, the volume occupied by the samples after tapping the measuring cylinder 100 times was noted and used to compute the tapped density [g/mL]. Triplicate determinations were made for each parameter and the average value was computed accordingly.
Carr’s compressibility index (CI)
and Hausner ratio (HR)
These were computed with data obtained from the bulk and tapped densities using the equations below (Equation 4):
True density
The liquid displacement method was adopted and liquid paraffin was used as the displacement fluid. Three determinations were made and the true density (Trd) was computed as shown below, where Wp is the weight of starch powder, x is the weight of the bottle and fluid, y is the weight of the bottle, fluid and starch powder, and SG is the specific gravity of liquid paraffin (0.865 g/mL) (Equation 6).
Determination of pasting properties
The pasting properties of the starches (NMNS and NMPS) were determined using a Rapid Visco-Analyzer (RVA; Perten Instruments, Macquarie Park, Australia). The method used previously by Shevkani et al.13 was adopted, but with slight modifications. Starch–water suspensions based on a dry starch basis of 25 g were used in monitoring the viscograms of the starches. The different batches of suspensions containing NMNS and NMPS were tested under similar temperature and time conditions.
Results and Discussion
The physicochemical properties of NMNS and NMPS are displayed in Table 1. The angle of repose is a reflection of the flow ability of a material and values <30° indicate excellent flow, those between 31° and 35° show good flow, while values between 36° and 40° and >40° signify that the material exhibits fair and poor flow, respectively.14 This indirect measurement of the flow properties of the starches show that NMNS and NMPS had angles of repose of 38.60° and 33.69°, respectively (Table 1), and indicate that pre-gelatinization improved flow of the starch powder.
The ability of both starches to deform under pressure was computed using CI. Materials with a CI ≤ 10% are considered to have excellent flow, those between 11% and 15% − good flow, between 16% and 20% − fair, and those >25% − poor flow. On the other hand, the measure of material cohesiveness, which depicts the degree of densification, was determined with the HR. Values ≤1.11 show that the material is cohesive, and it is less cohesive when HR values are between 1.12 and 1.20.15 The CI of NMNS was found to be 10.97% and that of NMPS was 7.50%, while the HR was 1.12 and 1.05, respectively. These results indicate that both starches exhibit good flow. However, the process of pre-gelatinization improved the flow characteristics of NMPS appreciably. The Trd of NMPS (1.39) was higher than that of NMNS (0.89), suggesting an increased ability of NMNS to promote even packing when confined into processing spaces such as compaction or tableting dies.16 The ability of materials to flow is dependent on the particle size, shape and distribution of the particles in the material. Disruption of the starch granules and consequent agglomeration due to gelatinization (Figure 1) produces voids within and between the particles. This reduces the friction between particles and allows for better flow, which invariably influences the packing behavior of the starches. Adequate/good flow is highly recommended in high-speed tableting/capsule filling machines as this ensures that the die cavities are uniformly filled, giving rise to tablets/capsules of uniform weight and uniform content.
The pH of the starches was 7.00 and 6.04 for NMNS and NMPS, respectively (Table 1), and falls with the specification of between 4.5 and 7.0 for starch solutions.17 The near-neutral pH of both starches implies that problems with irritability in the gastrointestinal tract might not arise from the use of these starches, and therefore, they would be desirable for use in oral formulations.18 The lower pH of NMPS observed in this study, which may be attributed to the modification process, is in agreement with a previous report by Azubuike et al.11 that examined pre-gelatinized Borassus aethipom starch.
The HC signifies the total amount of water that is retained by starch gel under defined conditions like pressure or heat.19 Pre-gelatinized starch was observed to have the ability to retain twice the weight of water retained by NMNS (Table 1), and the HC of NMNS was 25.80% while that of NMPS was 59.00%. The increase observed in NMPS can be attributed to the loss of crystalline association in the granules, leading to more available binding sites for water.20 The process of pre-gelatinization reduces the molecular weight of starch granules resulting in the ability to attract more water molecules and swell as compared to un-gelatinized starches.21 Materials with high HCs such as NMPS have been documented to be useful as disintegrants because of their increased ability to absorb water and swell, which is part of the mechanism of action of starch as a disintegrant.22
Swelling is an important characteristic that reveals the ability of starch granules to absorb water. Investigating the swelling behavior of starch within different temperature ranges is important as it evaluates the performance of a starch under industrial conditions. Table 2 shows that swelling power of both starches increased with an increase in temperature up to 80°C, with more pronounced swelling occurring at higher temperatures (70°C and 80°C) than within lower temperature ranges (between 40°C and 60°C). Increased swelling at higher temperatures has been ascribed to a weakening of the intrinsic binding forces in the amorphous regions of the starch granules at varying temperatures.23 Swelling was also found to be higher between 40°C and 60°C in NMPS than in NMNS. This effect can be ascribed to the destruction of the regular structure of the NMPS during pre-gelatinization, which conferred on it the ability to imbibe water increased over the temperatures. Swelling was observed to decrease considerably at 90°C to 75.50% and 87.10% for NMPS and NMNS, respectively, and can be attributed to total loss of granule structure and rupture of the granules.24 This suggests that the maximum swelling temperature for both starches is 80°C, and can serve as a guide in determining where and how these starches (NMNS and NMPS) can be used.
Microscopic observation of the starches using SEM revealed that NMNS is discrete, smooth, non-porous, and almost spherical in shape (Figure 1A), while the shape of NMPS is non-discrete, gel-like, rough, and irregular (Figure 1B). Granules of pre-gelatinized starch (Figure 1B) were also observed to be aggregated, which is due to granular disintegration and the subsequent release of soluble components during the thermal process of gelatinization. The particle sizes measured with SEM were 16.5 mm and 5.9 mm for NMNS and NMPS, respectively. Heating starch has been postulated to affect swelling and rupture of starch granules, which invariably affects the size of the resulting starch product.25 Furthermore, the heating process may have caused the loss of the amylopectin crystalline region, with the consequent rearrangement of bonds within the granules leading to breakdown, distortion and loss of granule integrity. These observations are in line with similar studies showing the effects of pre-gelatinization on starch morphology.26, 27
The FTIR spectra of NMNS and NMPS scanned in the 4000–400 cm−1 range are displayed in Figure 2 and 3, while the absorption peaks and corresponding peak heights for NMNS and NMPS are presented in Table 3 and Table 4, respectively.
Broad bands observed in both spectra at about 3800 cm−1 and 3000 cm−1 correspond to O–H stretching of hydrogen bonded hydroxyl groups, which may be ascribed to the presence of intra- and intermolecular hydrogen bonds in the starch granules.28 The spectra of NMPS (Figure 3) shows a stronger, broader O–H stretch at about 3800 cm−1 with a peak height of 17.29 (Table 4) that is about 3 times higher than that of NMNS (6.88; Table 3). This can be attributed to the higher swelling capacity observed for NMPS. Vibrations at 3000 cm−1 and 2800 cm−1 are characteristic of C–H stretches associated with ring hydrogen atoms, and this vibration was observed to be intense in the spectra of NMPS. Previous studies have shown that such changes in modified starches are attributed to changes in the ratio of amylose to amylopectin, with a consequent influence on the physicochemical properties of the starch.29
Vibrations at about 2300 cm−1, which represent C–H stretch for the alkane group of compounds, were found to be similar for both starches (Table 3, Table 4). Absorption peaks at about 1930 cm−1 (Figure 2, Figure 3) correspond to scissors vibrations of O–H bonds as a result of water of hydration in the starches. However, vibrations in NMPS were observed to be stronger as evidenced by a sharper peak. Vibrations at about 1460 cm−1 are related to C–H bending and both peaks were observed to be more intense in NMPS than NMNS. Vibrations at about 930 cm−1 are the skeletal absorptions of α-1,4 glycosidic linkage characteristic of all polysaccharides, consisting of COH and C–O–C glycosidic stretching and bending.30 These vibrations were observed at the same absorption wave number for both starches and depict similarity in the basic fingerprint of both starches. The last region has been defined as the characteristic fingerprint of materials like starches.31
The results from FTIR spectroscopy show that pre-gelatinization did not introduce any new functional groups. However, this process affected the arrangement of the molecules in the starch particles as evidenced by the shifting of some of the bands.
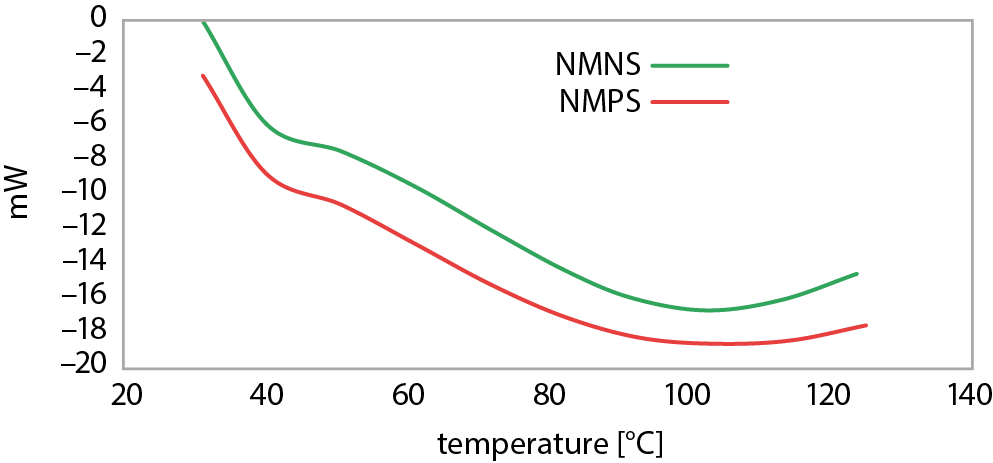
The behavior of NMNS and NMPS when subjected to heat was determined with DSC and the thermograms are displayed in Figure 4. The thermal properties of the starches such as onset temperature, peak temperature, conclusion temperature, and gelatinization enthalpy, among others, are presented in Table 5.
Although starch granules are insoluble in cold water, heat treatment in the presence of water causes gelatinization, which involves swelling of the granules, melting of the crystallite double helices, leaching of amylose, and consequent disintegration of the granules.32 This transitional phase of starch granules is a distinguishing property of individual starches that allows for their application in industrial processes.
Enthalpy of gelatinization (ΔH) gives an inclusive measure of the quantity and quality of crystallinity in starch granules, and is an indicator of the loss of molecular order within the granule that occurs with gelatinization.33 In other words, ΔH is related to the energy required to disrupt starch granules and is a reflection of the heat involved in breaking the bonds between starch granules. The outcome of gelatinization is influenced by shape, amylopectin chain length and crystalline portions of the starch.34
Table 5 shows that NMPS has a lower ΔH (5.27) compared to NMNS (6.38). This could be attributed to the fact that some double helices in NMPS may have been disrupted during the process of pre-gelatinization. Therefore, lower energy was required to initiate the gelatinization process of the starch granule. Furthermore, weakening of the starch granules during pre-gelatinization could have resulted in early rupture of the amylopectin helices, leading to the observed lower values for onset temperature (To), conclusion temperature (Tc) and peak temperature (Tp). Similar results have also been reported regarding some other modified starches.35, 36
In addition, since ΔH reflects melting of the crystalline region of the starch granules, the low values observed for NMPS may also be connected with low crystalline association within starch granules, indicating that less crystalline portions are present.37, 38 The degree of crystallinity of starch is known to be directly related to its granule strength, and demonstrates the extent to which the bond order within the starch molecule is broken and melted during the heating process.39 High ∆H shows that higher energy is required to disrupt the bond order because of the presence of strong bonds between the granules. However, a low ∆H, as observed for NMPS, shows that lower energy was required to break the bonds due to the rupture of the starch granules during the process of gelatinization.
The application of heat in an ordered manner and the analysis of the obtained viscograms are the measures that enable determining the pasting properties. When heat is applied to starch, there is a transformation of the granules from an initial ordered state to a randomized, but disordered state, which is a result of the swelling of the granules.40 Starch granules are usually saturated with water, but as heat is applied, the granules begin to swell and the water that has been imbibed in the granule aids melting of the crystal lattice of the starch, thus creating room for rapid movement of water between the granules and within the granules. Subsequently, more water molecules bind to the starch granules, while the swelling reduces the available water, resulting in physical interactions between the granules. These interactions lead to changes in the viscosity of the starch.
The pasting profiles and properties (derived from the viscograms) of NMNS and NMPS are shown in Figure 5 and Table 6, Table respectively. The pasting properties varied significantly among the starch samples. Native starch had better peak viscosity, breakdown viscosity and pasting temperature, while NMPS presented better trough viscosity, final viscosity, setback viscosity, and pasting time.
The high peak viscosity observed for NMNS (745.33 cP) may be due to the nonporous nature of the NMNS granules, leading to a greater volume of water within the crystal lattice and, consequently, a higher viscosity compared to the NMPS granules. Degradation of starch granules as a result of pre-gelatinization is responsible for reduced viscosity against shear stress,41 as observed in NMPS. The measure of peak viscosity, which is associated with the rate at which granule swelling equals granule breakdown, is slower for NMNS than for NMPS based on the aggregation of granules, as observed from the morphology using SEM. Granules of NMPS were more compact than those of NMNS and could account for the higher trough viscosity observed for NMPS. Trough viscosity reflects paste viscosity due to the disruption of starch granules upon heating and was observed to be higher in NMPS than NMNS as a result of the compactness of the granules.
Breakdown viscosity is a measure of the degree of disintegration of gelatinized starch granules during heating.42 The already gelatinized NMPS is thus expected to have a lower breakdown viscosity than NMNS due to the reduction in starch granules available for further disintegration to take place. The final viscosity indicates the stability of starch pastes to heat.43 The results show that NMPS exhibited a higher final viscosity (689.75 cP) than NMNS (360.75 cP), indicating that pastes from NMPS are more stable to heat than those from NMNS. In addition, final viscosity reflects the resistance of the starch paste to flow. The lower value observed for NMNS suggests its flow ease, which could be a result of more water molecules available within the crystal lattice of NMNS than in NMPS, while NMPS exhibited a higher resistance to shear and flow. Such high final viscosity and low breakdown viscosity properties have been described as desirable for many food and industrial processes.44
The setback viscosity is exhibited due to recrystallization of the amylose molecules in the gel45 and shows the tendency for retrogradation to occur. Although NMPS, which has a higher resistance to flow, would be expected to exhibit low setback viscosity because of the disruption of amylose molecules that took place during the pre-gelatinization process, the setback viscosity of NMPS (281.92 cP) was higher than that of NMNS (110.67 cP). This could suggest the tendency of NMPS to undergo some form of gelling upon cooling, a phenomenon that has also been demonstrated when pre-gelatinized cassava and rice starch were blended together.46 The temperature at which the viscosity of the starch material begins to increase during the heating process is known as the pasting temperature. According to Salman et al.,47 high pasting temperature is an indication of a high degree of crystallinity of starch molecules. The crystal lattice in the NMNS was more ordered than in the NMPS, which was disoriented due to pre-gelatinization, thus accounting for NMNS having a slightly higher pasting temperatures. However, the pasting temperatures of NMNS and NMPS were not significantly different. Visco-analysis shows that pre-gelatinization changed the pasting properties of native starch extracted from the tubers of N. mitis.
Conclusions
In this study, we have been able to modify starch from an underutilized crop, N. mitis tubers, by pre-gelatinization. Pre-gelatinization improved the flow properties, and increased the absorption capacity and SP, as observed with other pre-gelatinized starches. However, the granule strength was found to decrease. The pre-gelatinized N. mitis starch also exhibited peculiar pasting behavior with low viscosities but good thermal stability, as has been reported for other pre-gelatinized starches. The findings of the present study show that pre-gelatinized N. mitis starch may be applicable for food, cosmetics and pharmaceutical products where high swelling/hydration, low viscosity and low bond strength are required. However, further research to determine crystallinity and the molecular weight of the native and pre-gelatinized starches may be conducted.