Abstract
Poly(glycerol sebacate) (PGS) is an aliphatic polyester which attracted significant scientific attention in recent years due to its vast potential in biomedical applications with regard to tissue engineering. It has been presented in the literature in the form of 2D films, porous scaffolds or nonwovens, to name just a few. Moreover, various applications have been proposed as a component of composite materials or polymer blends. Its physicochemical properties can be significantly adjusted by means of synthesis and post-synthetic modifications, including cross-linking or chemical modification, such as copolymerization. Many scientists have discussed PGS as a new-generation polymer for biomedical applications. Its regenerative potential has been confirmed, in particular, in tissue engineering of soft tissues (including nerve, cartilage and cardiac tissues). Therefore, we must anticipate a growing importance of PGS in contemporary biomedical applications. This brief review aims to familiarize the readers with this relatively new polymeric material for tissue engineering applications.
Key words: tissue engineering, biomaterial, PGS, biomedical application, poly(glycerol sebacate)
Streszczenie
Poli(sebacynian glicerolu) (PGS) jest poliestrem alifatycznym, który w ostatnich latach skupił na sobie zainteresowanie wielu zespołów badawczych głównie ze względu na potencjał w zastosowaniach biomedycznych, w tym w inżynierii tkankowej. W literaturze jest opisywany, między innymi, w postaci dwuwymiarowych filmów, porowatych scaffoldów oraz włóknin. Ponadto, w niektórych przypadkach, naukowcy proponują wykorzystanie PGSu jako składnika kompozytów bądź mieszanin polimerowych. Właściwości fizykochemiczne poli(sebacynianu glicerolu) mogą być optymalizowane zarówno za pośrednictwem metody oraz parametrów syntezy, jak i poprzez modyfikacje post-syntetyczne takie jak sieciowanie, modyfikacja chemiczna lub kopolimeryzacja. Badacze przedstawiają PGS jako polimer nowej generacji do zastosowań biomedycznych. Jego potencjał regeneracyjny został potwierdzony w inżynierii tkanek miękkich (w tym tkanek nerwowych, chrzęstnych i sercowych). Jest więc kwestią czasu jego szerokie wykorzystanie we współczesnych rozwiązaniach z pogranicza biomedycyny. Ten artykuł przeglądowy stawia sobie za cel przybliżenie zastosowania poli(sebacynianu glicerolu) w inżynierii tkankowej.
Słowa kluczowe: inżynieria tkankowa, biomateriały, PGS, zastosowania biomedyczne, poli(sebacynian glicerolu)
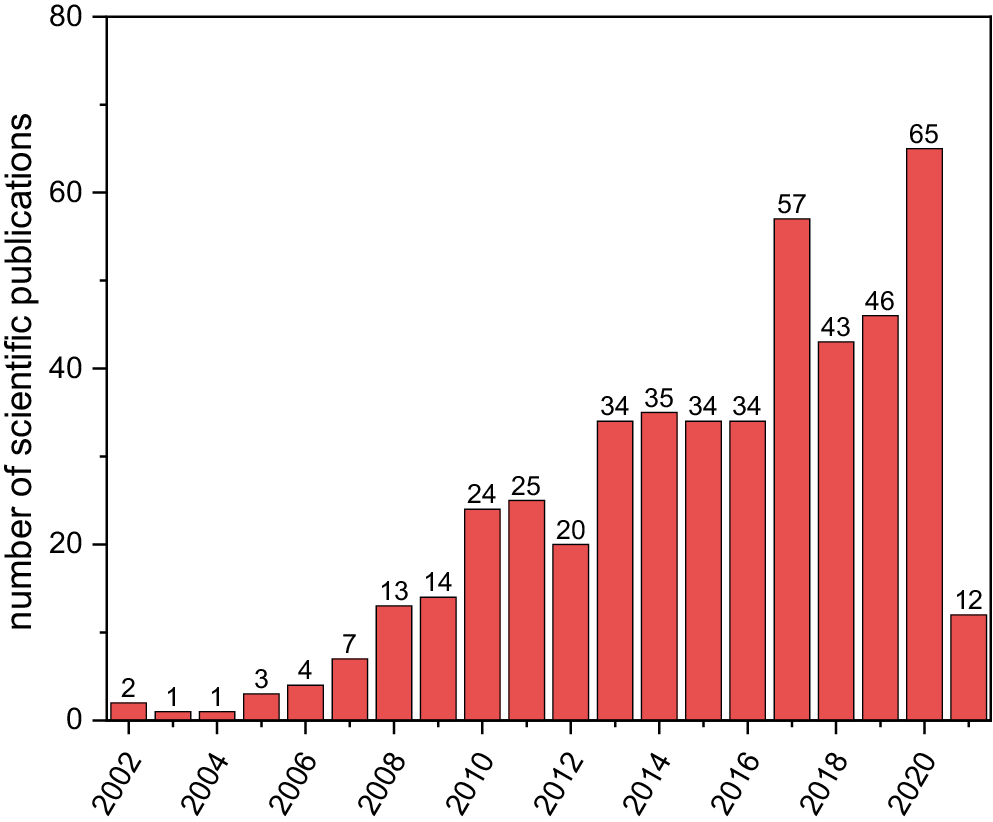
There are multiple biocompatible polyesters developed for biomedical applications including poly(L-lactide), polycaprolactone or polyglycolide.1 Among the aforementioned polymers, we can indicate a new emerging one: Poly(glycerol sebacate) (PGS). Poly(glycerol sebacate) is an excellent candidate for biomedical applications thanks to its physicochemical properties including its elastomeric capabilities,2 cross-linking potential,3 biodegrability4 and biocompatibility5 displayed in several in vitro and in vivo studies. Substantive research has been carried out on the field of PGS applications in various areas, especially biomedicine. The gradual increase in the annual number of papers regarding PGS is presented in Figure 1.
PGS: structure, synthesis, properties, and forming techniques
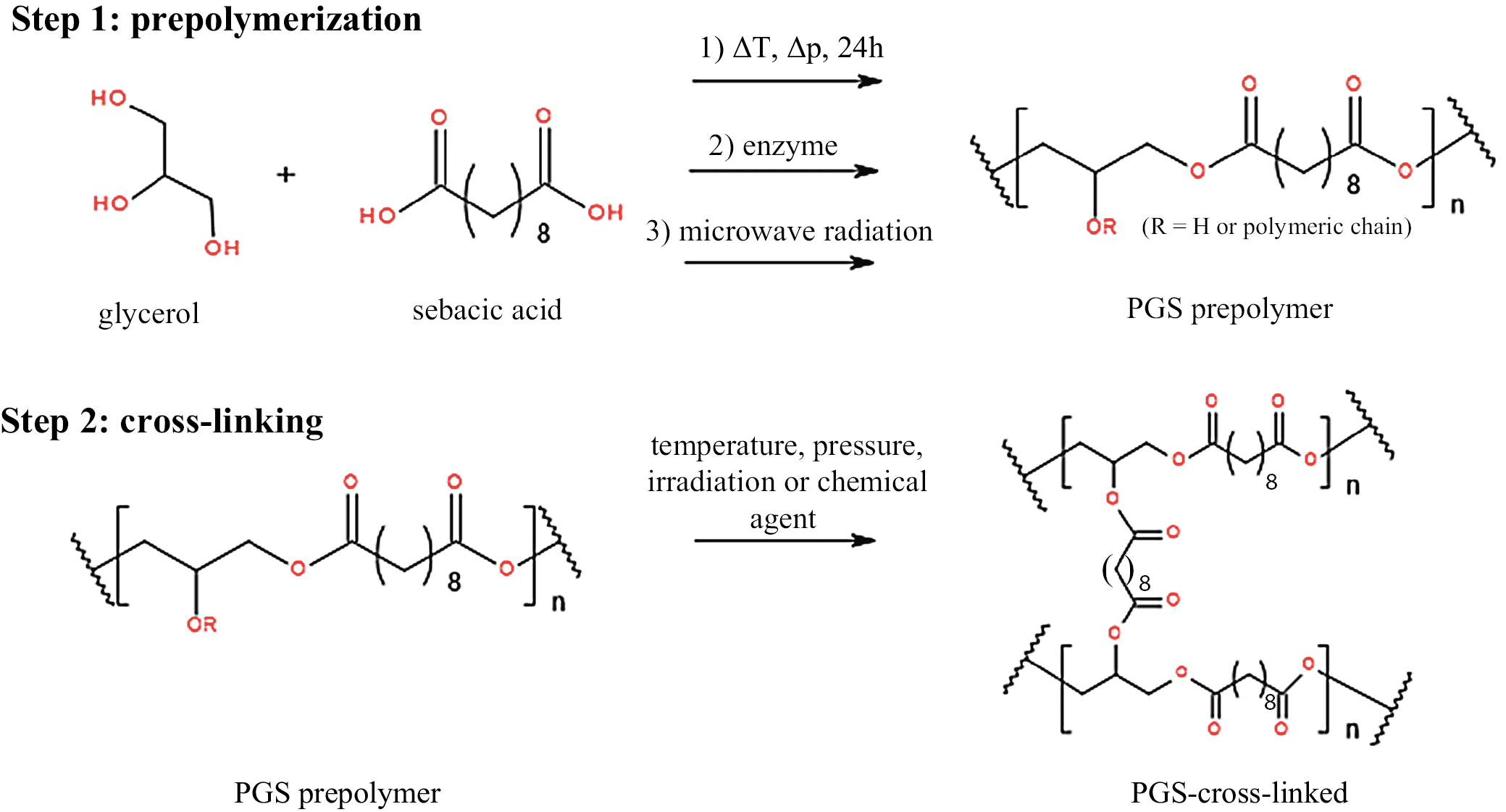
The PGS was first reported in terms of biomedical application by Wang et al.6 However, the described polymer was investigated in the aliphatic copolymers synthetic study by Nagata et al. in 1999.7 The PGS is an elastomeric, biodegradable polyester material, most commonly derived from dicarboxylic sebacic acid and glycerol through polycondensation.
Prepolymer synthesis
Commonly, PGS is obtained as a prepolymer (pPGS) which is subject to further structural modification, curing, blending, and forming. Each synthetic method results in different polymer structure. We can outline the most popular approaches in pPGS synthesis:
• reduced-pressure polycondensation,
• enzymatic synthesis and
• microwave-assisted polymerization.
Introduced in 2002,6 reduced-pressure polycondensation is undeniably the most trending synthetic pathway of obtaining pPGS.2, 4, 5, 8, 9, 10, 11, 12, 13, 14, 15 It consists of synthesis at elevated temperatures for a certain period of time in the inert gas atmosphere, combined with the subsequent reduction in pressure, most commonly to around 40 mTorr.
However, polycondensation in atmospheric pressure has been reported as well.7, 16, 17 The major difference in utilizing pressure is the propagation of inter-molecular bonding (i.e., cross-linking). Decreased pressure in combination with increased temperature in the 2nd stage of polymer synthesis results in the aforementioned cross-linked structure. Sometimes this stage may be delayed due to the process limitation (e.g., during porous scaffold formation cross-linking is postponed to the moment when scaffold is partially formed18).
Also noteworthy, reduced-pressure polycondensation is also mostly used as a technique for the synthesis of PGS copolymers (i.e., poly (glycerol-sebacate)-co-polyethylene glycol19 or poly(glycerol-sebacate)-co-polycaprolactone20).
In the enzymatic synthesis, the most commonly encountered catalyst is Candida antarctica lipase B (CALB), which is utilized to obtain PGS.21, 22 Perin and Felisberti reported a correlation between CALB selectivity and acyl migration phenomenon, which causes branching during the PGS polycondensation.22 The enzymatic synthesis is performed at slightly elevated temperature and has proven to produce polymer with increased linearity.21
Microwave radiation has also proven to shorten the prepolymer synthesis time before the cross-linking stage occurs.23 Moreover, it helps to obtain homogeneous polymer mixture or dispersion (e.g., when forming composites).3 The most commonly encountered approach consists of interval microwave irradiation (e.g., 1 min 650 W with 10 s interval24). In terms of backbone architecture, microwave synthesis can lead to more branched structures in comparison to conventional melt polycondensation.25
The described synthetic approaches are presented graphically as a synthetic pathway in Figure 2.
Cross-linking and branching
of the polymer chain
A thorough PGS branching characterization method by means of 1H NMR spectrum was introduced by Perin and Felisberti.22 This method allows for an easy and effective structural characterization of prepolymer before subsequent cross-linking or modification. In-depth structural analysis conducted by means of 1H NMR allows for an in-depth definition of the pPGS structure. Namely, one can outline the presence and abundance of specific glicerydic units in the polymeric chain and therefore, analyze the topology of the molecular chain.22, 26 This method can be utilized to determine the linearity of the obtained PGS by confirming the presence of 2 individual linear units. As described in the previous section, various synthetic approaches result in different branching degree, degree of esterification or simply degree of polymerization.
One can distinguish cross-linking techniques depending on the utilized agent. The most popular pathway to form intermolecular bonding is thermal cross-linking. It can be performed for a shorter period of time at higher temperature (taking into consideration degradability of PGS) or longer (even several days) in a lower range of temperatures (120–150°C).27, 28 The cross-linking temperature and time considerably influence the degree of cross-linking and thus, the mechanical properties of the polymer.
Chemical cross-linking of pPGS can take effect by compounds such as methylene diphenyl diisocyanate (MDI) or hexamethylene diisocyanate (HDI). They can be introduced to pPGS prior to the curing process in order to increase the cross-linking degree, tensile stress and Young’s modulus.13, 17
There is a broad array of structural modifications which introduce photocurable groups to the prepolymer (e.g., methacrylate groups12). Modified polymeric backbone can be further subjected to irradiation resulting in photocured structures. Therefore, we consider an irradiation as another agent capable of cross-linking poly(glycerol sebacate).12, 14, 26, 29
PGS polymer properties
Until now, PGS has been seen as a material that resembles soft tissues.17, 30 Thanks to the endogenous character of sebacic acid and glycerine,5, 6, 25 PGS and PGS-based composites are recognized as biocompatible.14, 31 The PGS is obtained as transparent or slightly yellow polyester (depending on the presence of oxygen during the reaction).
The structural analysis of the PGS performed using Fourier-transform infrared spectroscopy (FTIR) confirms the presence of all important bonds and functional groups, including polar hydroxyl, terminal carboxyl groups, ester bonding, and aliphatic backbone.27
In terms of physicochemical characteristics, PGS is considered an elastomeric material. Tests on tensile strength performed on PGS strips divulged an elastomeric character of the polymer by the presence of the characteristic stress-stain curve.6 According to literature reports, PGS exhibits Young’s modulus in the range of 0.12–0.5 MPa, a tensile strength of 0.27–0.50 N/m2, and an elongation at break of 180–274%.6, 23, 32 Moreover, the polymer is considered hydrophilic with a water contact angle around 70–80°.20, 33 At body temperature (~37°C: the temperature of potential biomedical applications) it is fully amorphous.6, 23
In addition, the prepolymer (pPGS) is soluble in many readily available organic solvents, such as 1,3-dioxolane, tetrahydrofuran, ethanol, isopropanol, and N,N-dimethylformamide, dioxane. This makes it easy to process using a variety of techniques.6
Processing techniques
of PGS and PGS-based materials
Poly(glycerol sebacate) has proven to be readily processable using various techniques. The simplest of them is to give it the suitable shape using in-mold polymerization combined with cross-linking.6, 27 However, in addition to the target shape, several other factors must be considered when designing the material for potential tissue engineering applications. These include porosity, mechanical properties and biocompatibility. When discussing PGS processing, one should also note that the pre-polymer form is mostly processed (in non-cross-linked form). Cross-linking is one of the last steps that stabilizes the properties of the product. Some of the most commonly used techniques for poly(glycerol sebacate) processing are described below.
Solvent casting
The solvent casting method is the most commonly used method of producing materials based on PGS. It consists of the following steps: 1) dissolving the polymer in a solvent (in the case of PGS – dissolving the non-cross-linked prepolymer); 2) pouring the solution into a prepared mold or vessel (e.g., Petri dish); and 3) evaporating the solvent, and cross-linking of the product.
In the case of PGS and PGS-based materials, the most commonly used solvents are dimethylformamide (DMF),13, 34, 35 tetrahydrofuran (THF)2, 5, 17 and dimethyl carbonate (DMC).36 The solvent casting method is often supported by the particulate leaching technique. The aim of this treatment is to obtain larger pores optimal for cell growth and development. For this purpose, solid particles are introduced into the polymer solution and washed out of the material after the cross-linking step. Lee et al. used 25–32-µm grounded salts as porogens, which after the production of the final element were washed out in water bath.14
The PGS-based materials made by solvent casting are very often subjected to a freeze-drying process in order to remove solvent residues from the structure.9, 37, 38 The solvent casting method has also been used to modify PGS by introducing a filler into the system.2, 35 For example, Gaharwar et al. introduced 1% wt of carboxyl functionalized multi-walled CNTs into the solution of PGS (in THF). These authors proved that, thanks to interaction between the filler and polymer matrix, the obtained material is characterized by much better mechanical parameters and resistance to degradation than unmodified PGS.2
Thermally induced phase separation method
The thermally induced phase separation (TIPS) method is based on the induction of phase separation in the liquid-liquid or liquid-solid system.39 The standard PGS manufacturing process using this method can be divided into 4 stages: 1) dissolving the prepolymer in a solvent (e.g., dioxane40); 2) freezing the system; 3) freeze drying – sublimation under reduced pressure of the polymer-lean phase (solvent); and 4) cross-linking. The product obtained using the TIPS method, after curing, takes the form of an elastic foam. An example is shown in Figure 3.
The TIPS method is used, i.a., to produce chemically cross-linked PGSU.40, 41 Hexamethylene diisocyanate (HDI) is used as a cross-linking agent. It reacts with the hydroxyl groups of PGS to form a chemically cross-linked poly(glycerol sebacate urethane) (PGSU).
Particulate leaching
The particulate leaching technique is most often used in parallel with other methods of PGS-based material fabrication. The use of solid porogens with specific geometry and particle size allows for the strict control of porosity of the biomaterial. This is important primarily for proper cell development. The size of the pores is also a very important factor in determining the ability to infiltrate cells deep into the scaffold.
Two approaches are mainly used in the production of PGS-based materials using the particulate leaching technique. The 1st of them is described in more detail above. It is based on the solvent casting method.9, 37, 42 It consists of the following steps: 1) filling the mold with solid porogen particles; 2) flooding the porogen bed with PGS and solvent solution; 3) evaporating the solvent and cross-linking the product; and 4) washing out the porogen. The 2nd frequently used approach is to mix the prepolymer directly with the porogen particles without the use of a solvent.31, 43 The rest of the procedure is similar.
The most commonly used porogens are sodium chloride particles (NaCl).9, 36, 42 Depending on the size of the target pores, the salt is ground and filtered prior to the process. Pashneh-Tala et al. have previously used sucrose as a porogen.11 The advantage of both compounds is their good solubility in water, and thus, the possibility of non-invasive removal of solid particles from the system using just distilled or deionized water (H2O) baths.36, 42 In addition to water, ethanol is used to wash the final product in order to remove unwanted post-process residues (e.g., residues of a cross-linking agent or oligomer fractions).44
Electrospinning
Electrospinning can be defined as a spinning fiber forming process based on an electrohydrodynamic phenomenon that uses electrostatic force to draw continuous fibers in the form of a liquid jet, from a polymer solution or melt.45 Typically, the product made using this method is in the form of a nonwoven mat of randomly ordered fibers.
When discussing the processing of PGS by electrospinning, it should be noted that the form of the prepolymer is processed. As with other methods, cross-linking is carried out only in the final stage of material production.
Scientific reports show that it is practically impossible to process PGS itself using the electrospinning technique. The main reason for this is its low molecular weight, resulting in the absence of entanglements.46, 47 This problem can be solved in several ways. One example is the electrospinning of PGS with a carrier polymer (e.g., PVA48), which is removed from the system after the process (by leaching). Another interesting approach is the electrospinning of PGS as a biocomponent in a blend with another polymer (i.e., poly(lactic acid) (PLA)46, 47 polycaprolactone (PCL)49 or poly(methyl methacrylate) (PMMA)50).
An important aspect is the selection of an appropriate solvent for the preparation of the polymer solution. Fakhrali et al. produced PGS/PCL fibers using various solvents (i.e., acetic acid, acetone, N,N-dimethylformamide, formic acid).49 The studies have shown that the selection of an appropriate solvent has an impact on the morphology of the fibers and their diameter.
3D printing methods
Three-dimensional printing includes a group of computer-assisted techniques to enable the production of 3D models. The starting material is applied to the work area in a layer-by-layer manner and solidified using various physical factors.
The literature presents a handful of approaches to 3D printing of PGS-based materials. Singh et al. used the stereolithography method to manufacture the Nerve Guidance Conduits, taking into account the ability to cross-link PGS with UV radiation.13 A similar approach was used by Wang et al., who utilized digital-light-processing-based 3D printing for the production of nature-inspired double network (DN) from poly(glycerol sebacate) acrylate (PGSA).29 A completely different method was proposed by Yeh et al.,who used extrusion-based 3D printing of acrylated poly(glycerol sebacate) (Acr-PGS) to manufacture the scaffold with elastic properties.51
Others
In addition to the methods described above, several other methods of fabrication and processing of PGS-based materials have been developed. These include, i.a., gyrospinning52 and micromoulding53 techniques. Such broad processing possibilities allow for a more complete use of the PGS application potential.
Degradation and biodegradation
The degradation behavior of PGS is a desirable characteristic for tissue engineering applications which aim for stimulating cell proliferation with the goal of eliminating the scaffold from the organism. The time needed for the complete dissolution of the PGS implant in the in vivo conditions is small and was determined to be 60 days.6 Its exact length depends on many factors. It was clearly shown by Pomerantseva et al. that curing time, which greatly affects cross-linking PGS degree, affects in vivo degradation time.4 Bulk materials need more time for decomposition in comparison to porous structures utilized as the cellular scaffolds. It naturally indicates the latter as potential candidates for bone tissue engineering applications in terms of biodegradation time.40 However, the degradation of the material caused by environmental factors such as oxygen or moisture applies to the biological systems as well. Therefore, in vitro and in vivo biodegradation studies of the PGS-based scaffolds are of vital importance on the field of tissue engineering applications.
Biocompatibility and cytotoxicity assessment
To consider any material for biomedical applications, its biocompatibility has to be proven and supported by relevant studies. Within the International Union of Pure and Applied Chemistry (IUPAC) definition, biocompatibility is an ability to be in contact with a living system without producing an adverse effect.54 Wang et al., while first reporting on biomedical applications of PGS, clearly took that property into consideration. Moreover, they noticed that the ideal scaffold for tissue implantation should be biodegradable and resemble properties of the extracellular matrix (ECM) in its physicochemical aspects.6 Bone defects after injuries, bone tumor resections or infections need to be supplemented by implants. The problem often faced in modern medicine and tissue engineering is to find a proper material that does not provoke rejection, is not limited in quantity and shape, and has specific mechanical properties similar to natural bone or even replaceable by one. If it is biodegradable and promotes bone development (i.e., material is osteoinductive), it can be replaced by a natural bone after application in humans. Often to fulfil those restrictive criteria, the implant material needs to be complex and modified.
Following this line of reasoning, many materials based on PGS with an admixture of other compounds (polymers, mineral apatites or others) are manufactured. For example, a study on the PGS/PCL blend of porous scaffolds for cartilage tissue engineering was conducted and showed successful results in terms of biocompatibility.55 The in vitro degradation time, adhesion and proliferation abilities were investigated on bone marrow-derived mesenchymal stem cells (BMSCs) and articular chondrocytes (ACCs). The weight loss rate of PGS/PCL scaffolds was significantly slower than that of pure PGS stent, which shows that PCL was responsible for prolongation of the degradation time. Moreover, PCL helped increase stiffness (evaluated with uniaxial mechanical test). Synthesized PGS/PCL material was analyzed using scanning electron microscope (SEM), as well as BMSC and ACC cell distribution. Cell morphology detection was observed using confocal microscope. As a result, authors obtained a biocompatible, composite material with favorable mechanical strength and appropriate biodegradability. Materials combining poly(glycerol sebacate) with polycaprolactone have also been studied by other scientists, showing a great potential for soft tissue engineering.56, 57 This brief review of the biological evaluation conducted on PGS-based materials is presented in the Table 1.
The PGS-based systems for tissue engineering constitute a diverse range of potential applications supported by biological evaluation. It positions PGS as a candidate for future-generation biomaterial suitable for a variety of biomedical purposes.
Conclusions
Poly(glycerol sebacate) is an emerging polymeric material for biomedical applications. With plenty of synthetic pathways followed by multiple cross-linking possibilities, the material proves to be a contestant for contemporarily utilized polymers for biomedical applications like PCL or PLA. The wide spectrum of applicable forming techniques broadens the possibilities of implementing this particularly interesting polyester in regenerative medicine. One has to take into consideration its drawbacks including low molecular weight distribution of prepolymer or its high rate of biodegradability. However, PGS displays the properties that modern regenerative medicine is currently searching for.