Abstract
Background. The pH of the skin surface is usually between 5.4 and 5.9 and functions as a barrier against bacteria and fungi; thus, the composition of the topically applied drug form may be of high importance for proper medication.
Objectives. To evaluate the influence of the measurement conditions in aqueous solutions of ointments, creams, and gels, which include polymeric components, on the pH and conductivity results.
Materials and methods. The pH and electrolytic conductivity of aqueous dispersions of commercially available ointments, creams and gels were tested and compared to reference vehicles.
Results. The results of the dilution method measurements of the pH and electrolytic conductivity of the ointment preparations are highly diverse, ranging from 5.88 to 6.27, whereas the reference pH for Unguentum simplex was between 5.40 and 5.43. Furthermore, the measurements of the pH and electrolytic conductivity with the dilution method for creams did not provide repeatable results with a small sample size, and the pH of commercial preparations was in the range between 5.79 and 6.37, compared to the reference pH of 5.23–5.46. However, the dilution method for measurements of the pH and electrolytic conductivity was suitable for hydrogel preparations and the obtained results were repeatable in the range of 6.11–6.90, while the reference preparations were in the range of 5.19–5.62.
Conclusions. Evaluation methods of the electrolytic conductivity and pH of the preparations applied on the skin should be further evaluated; however, the pH of the commercial preparation seems to differ from the physiological skin pH, which covers the range of reference preparations.
Key words: po----lymer, pH, electrical conductivity, gel, ointment
Streszczenie
Wprowadzenie. Odczyn na powierzchni skóry wynosi zwykle 5.4–5.9. Wartość ta wpływa korzystnie na funkcje barierowe skóry wobec bakterii i grzybów. W konsekwencji skład postaci leku stosowanej miejscowo na skórę może mieć duże znaczenie dla wyników terapii.
Cel pracy. Ocena wpływu warunków wykonywania pomiarów w wodnych rozproszeniach maści, kremów i żeli, zawierających składniki polimerowe, na wyniki pH i przewodnictwa.
Materiał i metody. Zbadano pH i przewodnictwo elektrolityczne wodnych dyspersji maści, kremów i żeli dostępnych na rynku, oraz porównano te wartości z preparatami odniesienia.
Wyniki. Wyniki pomiarów pH i przewodności elektrolitycznej preparatów maści metodą rozcieńczania są bardzo zróżnicowane, w zakresie 5.88–6.27, podczas gdy referencyjne pH dla maści prostej wynosiło 5.40–5.43. Również pomiary pH i przewodności elektrolitycznej metodą rozcieńczania w przypadku kremów nie dają powtarzalnych wyników, kiedy stosuje się niewielką ilość próbek. Odczyn pH preparatów handlowych zawiera się w przedziale 5.79–6.37, w porównaniu do odniesienia 5.23–5.46. Metoda rozcieńczania pomiaru pH i przewodności elektrolitycznej wydaje się być odpowiednia dla preparatów hydrożelowych: uzyskane wyniki dla preparatów handlowych są powtarzalne w zakresie 6.11–6.90, a dla preparatów odniesienia w zakresie 5.19–5.62.
Wnioski. Metody oceny przewodności elektrolitycznej i pH preparatów podawanych miejscowo na skórę wymagają dalszej oceny, jednak pH preparatów handlowych wydaje się odbiegać od fizjologicznego pH skóry, które obejmuje zakres preparatów referencyjnych.
Słowa kluczowe: polimer, pH, przewodnictwo elektryczne, żel, maść
The pH of the skin surface is usually between 5.4 and 5.9, and provides a barrier function against bacteria and fungi.1 Medicinal substances are usually delivered to the skin in the form of an acid or base, rather than a salt, as far as the technological considerations allow. Consequently, the penetration of these drugs through the skin is a function of the dissociation constant (pKa), along with the pH of the preparation and the pH of the superficial layer of the skin. European Pharmacopoeia contains a chapter on physical testing, including a description of the method of pH aqueous solutions and a recommendation for samples to be diluted in distilled water.2 Typically, fatty formulations before measurement must be emulsified in distilled water. Hydrophilic gels and creams may be diluted before the measurements and the results are recorded after a certain time. The measurements of lipophilic ointments may be carried out in an aqueous extract obtained by extraction in hot water. Various polymers are applied to constitute the drug form, which may be crucial for the pH and conductivity conditions of the preparation. Moreover, lipophilic ointments often contain the long chain alkanes, which are classified usually behind the polymers, whereas creams and ointments are composed using classical macromolecules described as polymers. Many topically applied drugs are developed on the basis of non-ionic or ionic polymers – for example, methylcellulose or polyacrylic acid.
The applied pH tests are diversified. For aqueous solutions of ointments, different authors propose various modes of sample preparation. Popescu et al. mixed 2 g of ointment sample with 30 cm of water and 5 g of paraffin in a baker, heated the mixture in a steam bath for 30 min with occasional stirring, and then assessed the pH only after cooling and filtering the sample to remove paraffin.3 According to Rajasree et al., 1 g of the ointment was dissolved in 100 mL of distilled water, left for 2 h, and the pH was measured.4 Kenley et al. diluted the cream sample before measurement with distilled water at a ratio of 1:4.5 Kumar et al. postulate that the direct measurement of cream in the layer is 0.5 cm.6 Nesseem recommends the preparation of 1 g of cream in 30 mL of distilled water with a stable pH of 7.7 Aqueous gels show good performance in topical applications due to the hydrophilic nature of the polymers and high dispersibility in water. Some authors propose direct examination of the pH of the hydrophilic gels in undiluted samples.8 Nagaich et al. studied the pH of a 1% aqueous gel solution after its dissolution in 100 mL of distilled water and incubation for 2 h in standard conditions.9 Quiñones and Ghaly dissolved 0.3 g of gel in 100 mL of distilled water, protected the sample from light for 2 h, and then measured the pH.10 Other investigators have dissolved the gel in distilled water at a proportion of 10% by volume, and the pH measurement was performed in triplicate.11
Objectives
The aim of this study was to evaluate the influence of the measurement conditions in aqueous solutions of ointments, creams and gels, which include polymeric components, on the pH and conductivity results.
Materials and methods
Materials
The following ointments were used: 1) Unguentum simplex consisting of white petrolatum and anhydrous lanolin in a ratio of 90:10, prepared in-house using certified pharmacopoeial components (US); 2) a protective ointment with vitamin A containing 800 IU/g of retinol palmitate with white petrolatum and Palsgaard 0291 as an emulsifier (UA); 3) the zinc ointment containing zinc oxide on a base of hydrophilic petrolatum (UZ). The assessed creams included: 1) cream base Hascobaza with hydrocarbons, emulsifiers, and typical polar solvents (CH); 2) cream with 0.5% of hydrocortisone acetate with white petrolatum (CA); 3) 1% cream with clotrimazole (CC). The following aqueous gels were assessed: 1) 0.5% methylcellulose gel (GM); 2) gel with 100 mg/g of ibuprofen lysine salt, with macrogol and polyacrylic acid (GI); 3) gel with 8.5 mg/g of sodium heparin, using a base of neutralized acrylic acid polymer (GH).
Methods
Aqueous dispersions of the ointments were prepared using 5 g of the ointment and the addition of 45 mL of distilled water. The closed flask was then heated in a water bath at 70°C with frequent shaking. After macroscopic homogenization of the components, shaking was continued for another 5 min. Dispersions were then cooled and filtered through a medium porosity filter. Aqueous dispersions of creams and gels were produced with 5 g of formulation supplemented with 45 mL of distilled water, so that the weight to volume ratio of the mixture was 1:10.
The mixture was dispersed to obtain a homogenous suspension or solution. The aqueous solutions prepared this way, with the dilution of 1:10, were subject to potentiometric and conductometric measurements. During the measurements, the samples were diluted with distilled water to the value of 1:20, 1:30, 1:40, and 1:50, and were also studied.
The pH was tested using the pH-meter from Meratronik (type 517; Warszawa, Poland) with a combined electrode SAg P201 and temperature compensator Pt-100. Each sample test was performed 5 times, and 2 samples were evaluated in parallel. A stabilization time of 3 min was applied before every measurement.
The electrolytic conductivity tests were performed using the conductivity device CC-505 from Elmetron (Zabrze, Poland). Each sample measurement was performed 5 times, and 2 samples were performed in parallel. A stabilization time of 2 min was applied, and each test was conducted at room temperature.
Results
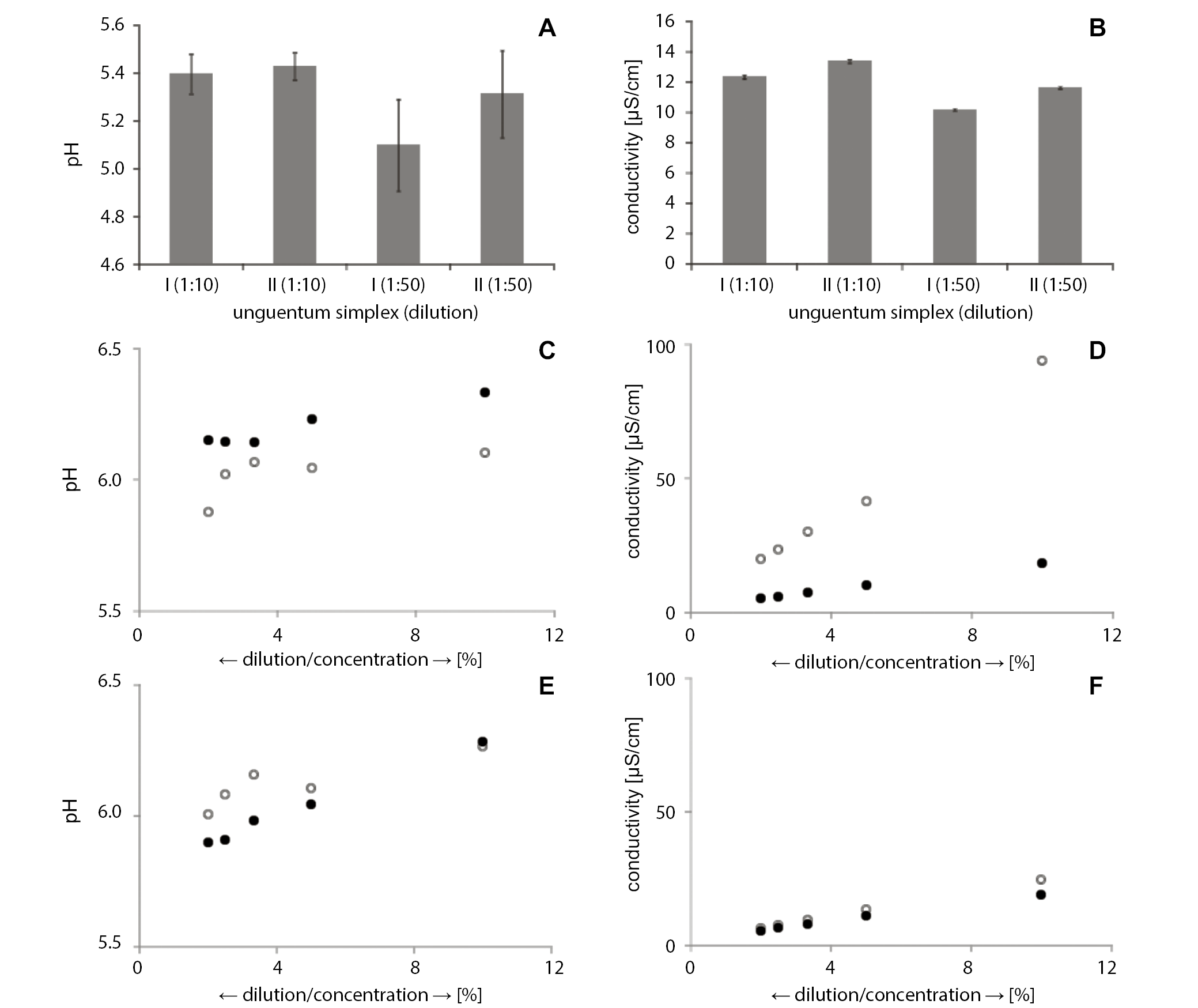
The pH and conductivity values of ointment-type preparations
As shown in Figure 1 in the column graph, the pH of the reference ointment base (US) with a dilution of 1:10 ranged from 5.40 to 5.43, while for the dilution of 1:50 it was between 5.10 and 5.31 (Figure 1A). The electrolytic conductivity of the US with the dilution of 1:10 was 12.35–13.39 µS/cm.
Figure 1C presents the pH of the ointment with vitamin A (UA) after dilution with distilled water at a ratio of 1:10 – it ranged between the values of 6.10 and 6.33. When we increased the dilution to 1:20, the pH slightly decreased to the range of 6.04–6.23. As we diluted the samples further (1:30, 1:40 and 1:50), the average pH value ranges were 6.07–6.14, 6.02–6.14 and 5.88–6.15, respectively. Interestingly, the lowest (1:10) and highest (1:50) dilutions showed a wide range of pH values in the investigation of the UA ointment, while the 1:30 dilution showed low variability. The Student’s t-test showed statistically significant differences of the pH measurements between the 2 samples performed at a 95% confidence interval (95% CI).
The average value of electrolytic conductivity of UA in the 1:10 dilution presented significant differences in both samples (I and II), ranging from 18.41 µS/cm to 94.06 µS/cm (Figure 1D). At the higher dilution of 1:20, the pH values decreased by almost 50% and ranged from 10.19 µS/cm to 41.44 µS/cm. In the 1:30, 1:40 and 1:50 dilutions of the ointment sample, the average conductivities decreased, and were, respectively, 7.28–30.16 µS/cm, 5.87–23.42 µS/cm and 5.14–19.85 µS/cm. The highest result, characterized by the greatest variation, was observed in the lowest dilution of 1:10. The remaining results were more uniform.
The average pH of the UZ at a dilution of 1:10 represented a narrow range of 6.27–6.28 (Figure 1E). The 1:20 dilution resulted in decreased values (6.04–6.11). This decrease was consistently shown in further dilutions, as the 1:50 dilution reduced the pH values to a range of 5.90–6.01. The decreasing pH values were characterized by a slightly increased variability.
The conductivity value of UZ in the 1st dilution with distilled water (1:10) was in the range of 19.00–24.62 µS/cm (Figure 1F). With higher dilutions – 1:20 and 1:30 – the values were, respectively, 11.01–13.37 µS/cm and 8.00–9.55 µS/cm. In further successive dilutions of UZ at ratios of 1:40 and 1:50, the average electrolytic conductivity values were similar – in the range of 6.49–7.52 µS/cm and 5.47–6.45 µS/cm, respectively. The Student’s t-test analysis did not show any significant statistical differences between the results of the electrolytic conductivity study using a 95% CI. Similar to the pH study of the UZ, the result that was most different from the rest was obtained with the lowest dilution (1:10). This result, however, was characterized by the largest spread, in comparison to the rest. The other results showed the downward trend, similar to the results identified in the pH values of UZ.
The pH and the conductivity
of oil/water (o/w) cream-type preparations
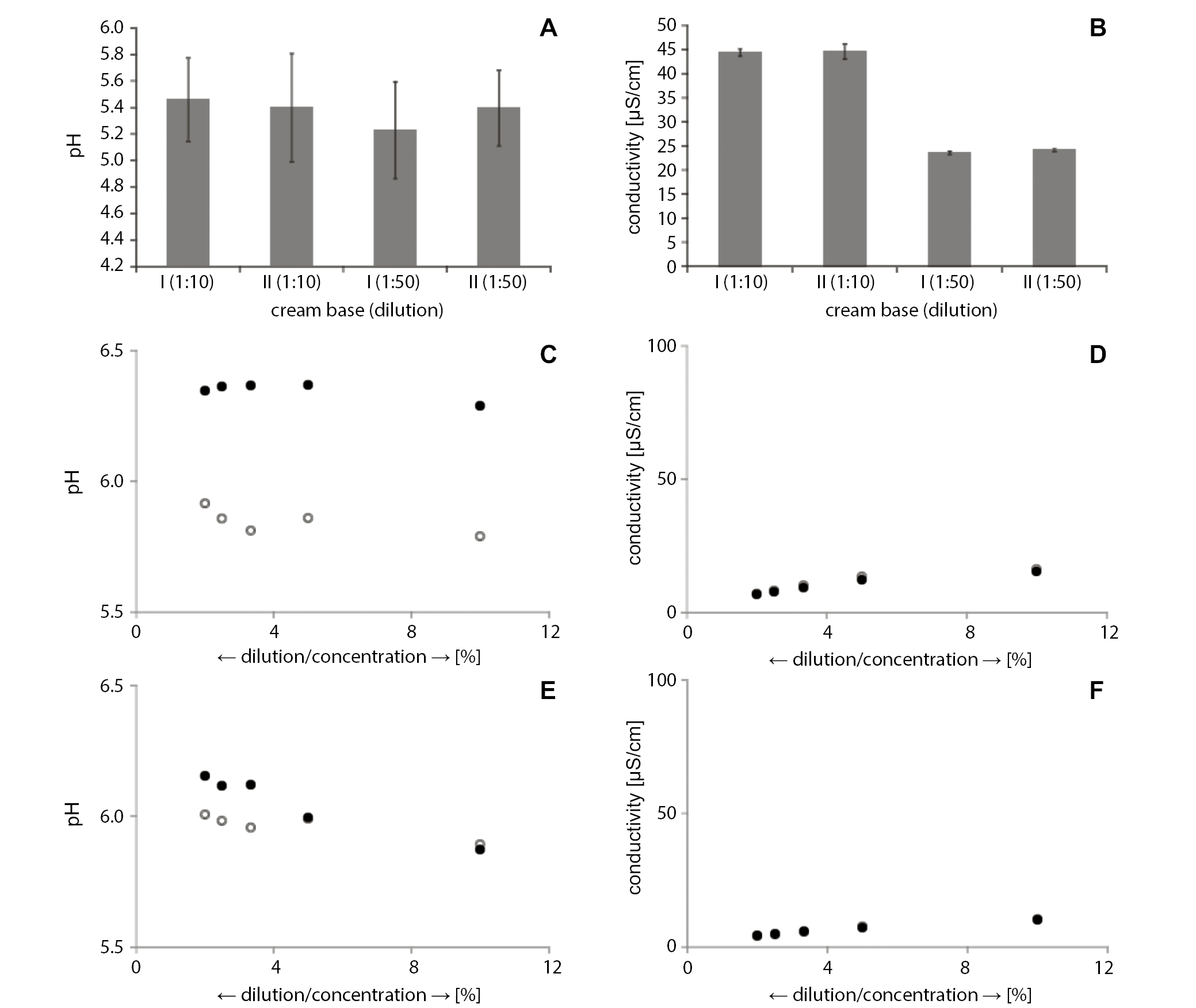
The pH of the model base of o/w cream CH was 5.23 and 5.46 for dilutions of 1:10 and 1:50, respectively (Figure 2A), whereas the electrolytic conductivity, as shown in Figure 2B, was in the range of 44.46–44.66 µS/cm and 23.62–24.24 µS/cm, respectively.
After diluting with distilled water at a ratio of 1:10, the average pH values of CC were fairly divergent at 5.79 and 6.29 (Figure 2C). With respect to the 1:20 sample dilution, values of both series slightly increased, respectively, to 5.86 and 6.37. In sample I, the average values of the pH obtained slightly increasing values along with the increasing dilution (1:30, 1:40 and 1:50); specifically, they were 5.81, 5.86 and 5.91, respectively. In sample II, we observed minimal reductions in pH following dilution – to 6.37, 6.36 and 6.35, respectively. The spread of the values was also similar, which may prove the lack of homogeneity of the studied cream. Moreover, the obtained results provided information on the repeatability of the study, regardless of the dilution ratio.
The average values of the electrolytic conductivity in both samples were similar at every stage of the study (Figure 2D). At the 1:10 dilution, the results were in the range of 15.46–16.24 µS/cm. At dilutions of 1:20, 1:30 and 1:40, the value of the measured parameter was in the range of 12.17–13.49 µS/cm, 9.42–10.06 µS/cm and 7.72–8.21 µS/cm, respectively. The highest diluted sample (1:50) displayed an average electrolytic conductivity value of 6.88–7.04 µS/cm. These results showed a similar downward trend that was seen above in present research. The broadest range of the obtained results was observed with the 1:10 and 1:20 dilutions, whereas the 1:30, 1:40 and 1:50 dilutions displayed a much narrower range.
The distribution in the test results of the pH of the aqueous solution of cream with hydrocortisone (CA) was similar in both samples. The 1:10 dilution pH values ranged between 5.87 and 5.89. Further dilution (1:20, 1:30, 1:40, and 1:50) of the CA led to values of 5.99, 5.96, 5.98, and 6.01, respectively. In sample II, the 1:30 and 1:40 dilutions exhibited pH values of 6.12, and reached 6.15 in the 1:50 dilution (Figure 2E). The analysis of the CA pH test results showed that, contrary to data from the studies of other preparations, the lowest pH values occurred in the 1st dilution of 1:10. In the other studied preparations, this value was the highest. These results showed a rising trend that significantly deviated from the results obtained in the 1st dilution. The values obtained for the dilutions of 1:30, 1:40 and 1:50 showed a rather large spread in comparison to the results obtained for the 1:10 and 1:20 dilutions.
The results of the conductivity measurements showed slight differences (Figure 2E). Initially (1:10), the conductivity was 10.03–10.26 µS/cm, and this decreased to 7.16–7.50 µS/cm in the 1:20 dilution. Further dilutions of the CA (1:30, 1:40 and 1:50) displayed the following results: 5.62–5.77 µS/cm, 4.60–4.86 µS/cm and 4.07–4.16 µS/cm, respectively (Figure 2F). Based on these data, we can conclude that conductivity values decreased in response to increasing dilutions with a relatively low amount of variability.
The pH and conductivity
of hydrogel-type preparations
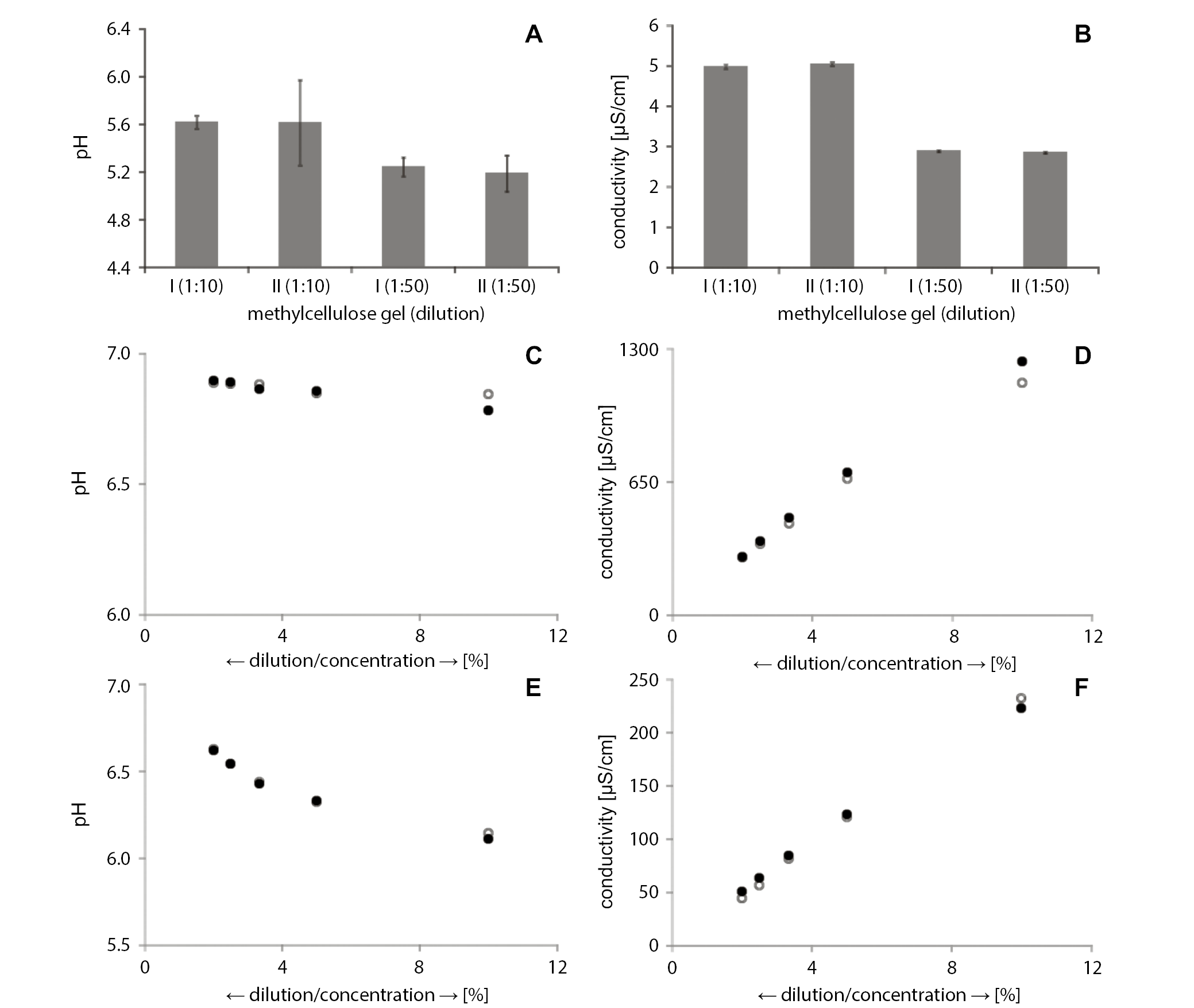
Results of the pH measurements of the dilution of reference preparation (GM) with methylcellulose 1:10 were in the range of 5.61–5.62, whereas after the highest dilution (1:50) the values ranged between 5.19 and 5.24 (Figure 3A). The electrolytic conductivity of the 1:10 dilution was in the range of 4.99–5.05 µS/cm, while it was in the range of 2.86–2.90 µS/cm in the 1:50 dilution (Figure 3B).
Average pH values of the gel preparation with 100 mg/g of ibuprofen lysine salt, with macrogol and polyacrylic acid (GI), ranged between 6.78 and 6.84 (Figure 3C). At dilutions of 1:20, 1:30, 1:40, and 1:50, the pH of the tested sample slightly increased and showed a rising trend: 6.85–6.86, 6.86–6.88, 6.88–6.89, and 6.89–6.90, respectively. The smallest value of the studied parameter was obtained at the 1:10 dilution, and this dilution displayed the largest range as well.
The average results of the five-time measurements of the electrolytic conductivity of GI indicated significantly higher values, in comparison to the previously analyzed data (Figure 3D). The results for the samples diluted 1:10 were in the range of 1133.80–1240.0 µS/cm. In further dilutions (1:20, 1:30, 1:40, and 1:50), electrolytic conductivity values decreased (666.40–695.80 µS/cm, 446.00–476.20 µS/cm, 348.00–361.80 µS/cm, and 281.00–283.60 µS/cm, respectively). A downward trend was observed following dilution, together with a range narrowing. The highest values were observed for the lowest (1:10) dilution.
The pH values for the gel preparation with heparin sodium salt (GH) increased on every step of dilution in both samples. This increase was significant, as each dilution resulted in a change of approx. 0.1 of a pH unit. In appropriate dilutions – 1:10, 1:20 and 1:30 – the pH values were in the range of 6.11–6.14, 6.32–6.33 and 6.43–6.44, respectively, while in the 1:40 and 1:50 dilutions, the average pH values were at 6.54 and 6.62 (Figure 3E). The lowest values of pH for the GH samples were observed in the 1:10 dilution, and further dilutions caused a rising trend. Overall, the span of the obtained results was narrow, and the highest values were observed in the distilled water dilution of 1:50.
The electrolytic conductivity data for GH are presented in Figure 3F. The 1:10 dilution values were in the range of 223.20–232.40 µS/cm, determined based on five-time measurements. With the increase in dilution to 1:20, the average results of the electrolytic conductivity decreased to the range of 120.60–123.40 µS/cm. In further dilutions, the average electrolytic conductivity value was in the range of, respectively, 81.40–84.40 µS/cm, 56.80–63.30 µS/cm and 44.60–51.00 µS/cm. The highest electrolytic conductivity value of GH was observed in the lowest dilution of 1:10, and these values decreased with subsequent dilutions. The narrowest span was shown by the values in the 1:20 dilution.
Discussion
In the study of the ointment, which analyzed separate samples for the same preparation, diversified pH and electrolytic conductivity results were clearly observed. In the preparation of the US, we observed a slight decrease in pH and electrolytic conductivity along with sample dilution. The greatest variability was observed in the UA. The wide changeability concerned both, the pH and the electrolytic conductivity. Due to the large amounts of lipophilic substances, UA hardly underwent dispersion in the aqueous environment. Numerous clumps of lipophilic substance were observed. In the case of UZ, we also noted high diversification of the results between successive samples in the pH measurements. Furthermore, a slightly smaller variability between sample measurements was observed in the study of electrolytic conductivity of UZ. This may result from different aqueous base numbers applied respectively in the UA and in the UZ. They are, respectively, 10 and 250. In the UA, white petrolatum was used with a low aqueous number, which does not favor particle hydrophilicity of the lipophilic substrate. The substrate in UZ, which contains the hydrophilic petrolatum with a high aqueous number, can be easily dispersed in water. No information was found on the influence of the Palsgaard 0291 used in the UA on the change of the aqueous number of petrolatum.
In the studies on the pH and conductivity of creams, we showed a clear difference between particular measurement series, particularly in the case of the ointment studies. In the case of CH, no pH changes were observed with dilution increasing from 1:10 to 1:50. Conductivity tests of this substrate showed that increased dilution significantly reduces its electrolytic conductivity. Particularly, large diversification between the measurement series was noted during the study of the pH in the case of CC; however, the diversity of results within one sample was small. This may reflect the heterogeneity of the parts of preparation squeezed from the tube, that were collected for testing. In the case of CA, a deviation of the obtained pH values was also present, but not to the same degree as observed in the ointments. The diversity of the results of the tested creams may be the outcome of their lipophilic nature. In studies on the electrolytic conductivity, significant changes were observed in the various dilutions. This may represent the so-called systematic error, which can result from the applied measurement method or other environmental influences. It should be emphasized that in the cream preparations, the diversity of the electrolytic conductivity results between the samples was smaller when compared to the ointment preparations.
In the gel preparation case studies, we noted a smaller diversity in the results. The values of the results were higher compared to those obtained for the ointment creams, and were nevertheless characterized by uniformity. For GM, the pH and electrolytic conductivity measurements displayed a clear decline with dilution. Results of the pH and electrolytic conductivity measurements for GI and GH showed slight differences, which can be categorized as a random error. These do not have a significant impact on the final measurement result, and their cause is unknown – likely a significant effect on the obtained results can be exerted by the composition of gel preparations, whose main component is water, while they contain little active substance and polymer.
Comparing the pH and conductivity changes in response to dilutions in the case of creams, we observed that the pH in particular samples was variable; however, the results of conductivity measurements were repeatable. Thus, further research should include conductivity as an auxiliary measurement allowing the quantitative assessment, comparable with the preparations of the cream type. The diversity of the pH values in the successive cream measurements, in the context of repeatable conductivity measurement results, requires further study. The formation of the balance between the concentration of ions connected with the emulsion system and the concentration of free ions in the solution likely occurs in the emulsion systems. This balance, however, does not reflect the concentrations of aqueous ions responsible for the pH.
Conclusions
The results of the dilution method measurements of the pH and electrolytic conductivity of the ointment preparations are highly diverse. Furthermore, the measurements of the pH and electrolytic conductivity using the dilution method in the case of creams do not give repeatable results when a small number of samples is used. Conversely, the dilution method for the measurement of the pH and the electrolytic conductivity is suitable for the hydrogel preparations based on polymeric materials, and the obtained results are repeatable. The methods of evaluation of the electrolytic conductivity and pH of the preparations applied on the skin should be further evaluated. The pH of the commercial preparations appears to be different from the physiological pH of the skin, which covers the range of reference preparations.