Abstract
Background. This study presents the results of an investigation into the interactions between innovative ophthalmic formulations and commercially available low-density polyethylene (LDPE) containers. The newly developed formulations are self-emulsifying oils (SEOs) containing suspended drug particles, designed to form an emulsion immediately upon contact with tear fluid. Physicochemical and mechanical properties of the containers were evaluated. Interactions between 6 different LDPE containers and the SEO matrix (oil “O” and surfactant Tween 20 “T”) were investigated, and the impact of the SEO formulations on the mechanical properties of the containers was assessed.
Objectives. The study aimed to identify potential interactions between SEO components and the packaging that may occur during storage under stress conditions, based on changes in morphology, structure, thermal behavior, and mechanical strength.
Materials and methods. The SEO carrier was prepared by mixing Miglyol® 812 with Tween® 20 at a concentration of 5% w/w, followed by sterile filtration. The suspensions were compounded aseptically using sterile, micronized sodium cefuroxime (CEF) and vancomycin hydrochloride (VAN) at a concentration of 5% w/w, along with sodium citrate (2% w/w). In accordance with stability testing guidelines, stress stability studies were conducted in a climatic chamber at 40°C/75% relative humidity and 60°C/75% relative humidity. To detect structural and physicochemical changes, advanced analytical techniques were employed, including Fourier-transform infrared (FTIR) and near-infrared (NIR) spectroscopy for the assessment of structural alterations and potential degradation, differential scanning calorimetry (DSC) for thermal analysis, and X-ray diffraction (XRD) for evaluation of material crystallinity. The mechanical strength of packaging material fragments after contact with the formulations was evaluated using a TA.XTplus texture analyzer.
Results. The experiments indicated potential migration (e.g., of plasticizers and residual monomers), as well as adsorption or absorption of excipient components. Subtle interactions were observed, accompanied by negligible changes in the mechanical strength of the packaging material.
Conclusions. The study confirmed the necessity of comprehensive compatibility testing between ophthalmic formulations and their packaging materials. A thorough understanding of these interactions is essential to ensure product stability, safety, and quality during storage and use.
Streszczenie
Wprowadzenie. Artykuł przedstawia wyniki badań interakcji innowacyjnych preparatów okulistycznych z komercyjnymi opakowaniami z polietylenu o niskiej gęstości (LDPE). Opracowane formuły olejowe mają postać samoemulgujących zawiesin olejowych (SEO), przeznaczonych do natychmiastowego tworzenia emulsji z płynem łzowym po aplikacji. Zaplanowano badania fizykochemiczne i stresowe opakowań. Badano interakcje 6 różnych opakowań z matrycą SEO (olej „O” i surfaktant Tween 20 „T”) oraz oceniono wpływ SEO na właściwości mechaniczne opakowań.
Cel pracy. Badania miały na celu identyfikację potencjalnych interakcji składników olejowego preparatu z opakowaniem, które mogą wystąpić podczas przechowywania preparatów, w oparciu o zmiany w materiale opakowania: morfologii, strukturze, własnościach termicznych i wytrzymałości mechanicznej.
Materiał i metody. Nośnik SEO przygotowano poprzez zmieszanie Miglyolu 812 z Tweenem 20 w stężeniu 5% w/w, a następnie poddano filtracji wyjaławiającej. Dodano mikronizowane proszki cefuroksymu sodowego (CEF) lub chlorowodorku wankomycyny (VAN) w stężeniu 5% w/w oraz cytrynian sodu (2% w/w), a następnie zhomogenizowano zawiesinę. Do badań przeznaczono próbki opakowań o wymiarach 5 × 5 mm, które umieszczano w probówkach Eppendorfa i zalewano 0,5 ml odpowiedniego płynu: oleju (O), oleju z Tween (O-T) lub zawiesiny (SEO-CEF, SEO-VAN).
Wyniki. Eksperymenty wskazały na możliwość migracji składników opakowania do preparatu (np. plastyfikatorów czy monomerów) oraz adsorpcji lub absorpcji do opakowania składników preparatu; w konsekwencji zanotowano również subtelne zmiany wytrzymałości opakowania.
Wnioski. Badanie dostarcza ważnych informacji, które muszą być uwzględnione przy wyborze opakowania dla kropli do oczu i wskazuje na przydatność użytych testów do oceny interakcji. Zrozumienie tych interakcji jest kluczowe dla zapewnienia stabilności, bezpieczeństwa i skuteczności terapeutycznej produktu podczas jego przechowywania i stosowania.
Key words: interactions, LDPE packaging, ophthalmic preparation, self-emulsifying oil
Słowa kluczowe: interakcje, opakowania LDPE, preparaty oftalmiczne, samoemulgujące krople olejowe
Introduction
One of the crucial steps in introducing a new medicinal product to the market is the development of appropriate packaging. Pharmaceutical packaging plays a key role in ensuring the quality, safety, and efficacy of the drug product. Its primary function is to protect the formulation from adverse external factors (physical, chemical, and microbiological), such as light, moisture, oxygen, temperature fluctuations, and contamination, which may lead to degradation of the active substance or deterioration of product properties. The container should ensure product stability throughout its shelf life. Furthermore, the packaging material must not interact with the formulation components.1
One of the most commonly used materials in pharmaceutical packaging, including packaging for ophthalmic products, is low-density polyethylene (LDPE), followed by high-density polyethylene (HDPE) and polypropylene.2, 3 These polymers are widely used due to their favorable mechanical properties, chemical resistance, and relatively low production costs. In ophthalmic formulations, appropriate packaging plays a critical role in maintaining sterility, stability, and therapeutic efficacy throughout the product’s shelf life. Ophthalmic solutions and suspensions are particularly sensitive to environmental factors such as light, oxygen, temperature fluctuations, and microbial contamination. Therefore, the selection of suitable packaging materials is essential to ensure patient safety and treatment efficacy.
Low-density polyethylene has a density ranging from
0.91 g/cm3 to 0.93 g/cm3. It is produced by radical poly-
merization of ethene under high temperature and pressure. The presence of side branches in the polymer chain prevents tight molecular packing, resulting in its relatively low density. This polymer is soft and resilient, resistant to puncture and tearing, and capable of withstanding repeated mechanical stress during use. These properties make it particularly suitable for squeeze bottles used in multidose ophthalmic products, where controlled dispensing and ease of handling are required. Although LDPE demonstrates good long-term stability, its resistance may significantly decrease under specific conditions, particularly following combined thermo- and photo-oxidative exposure.3, 4, 5 Furthermore, LDPE is chemically inert, exhibits good heat-sealing properties, and is readily processable, which facilitates large-scale manufacturing and ensures tight container closure.6 However, significant limitations of LDPE include low resistance to ultraviolet (UV) radiation and relatively high gas permeability.7 These characteristics may adversely affect the stability of light- and oxygen-sensitive active pharmaceutical ingredients (APIs), potentially leading to gradual degradation during storage.
Low-density polyethylene used in pharmaceutical containers may interact with drug products through several mechanisms, including adsorption (adhesion of drug molecules to the polymer surface, potentially leading to dose loss), absorption (penetration of the active ingredient into the polymer matrix), migration of container components (e.g., plasticizers or other additives into the formulation), photodegradation (degradation induced by UV exposure), and gas permeability (increasing the risk of oxidative degradation of formulation components).7 Disruption of the polymer structure may result in significant loss of active ingredients, potentially leading to reduced therapeutic efficacy.8 To mitigate these adverse effects, various material modification strategies have been developed. These strategies include the incorporation of colorants to improve UV protection, the addition of metal oxides or antioxidants to enhance barrier properties, and the application of surface coatings. Additionally, multilayer materials combining LDPE with other polymers or functional barrier layers are increasingly used to improve resistance to light, oxygen, and moisture. The appropriate selection and modification of packaging materials for ophthalmic products are essential to ensure drug stability, sterility, and therapeutic efficacy. Continuous advances in polymer engineering and multilayer technologies play a key role in meeting regulatory requirements and addressing the growing demand for high-quality, safe, and reliable ophthalmic drug delivery systems.
Objectives
The aim of this study was to investigate the effects of self-emulsifying oils (SEOs) and antibiotic suspensions formulated in SEOs on the structural and physicochemical properties of commercially available packaging materials. In addition, the influence of SEO components on spectroscopic, thermal, and mechanical properties was evaluated following stress testing. The development of new medicinal products requires the submission of extensive documentation in the Common Technical Document (CTD) format to the relevant regulatory authority, such as the Polish Office for Registration of Medicinal Products (URPL). The Quality module (Module 3) of the CTD requires the submission of stability data, including information on formulation–packaging interactions. Therefore, comprehensive physicochemical and stress testing of the packaging materials was incorporated into the study design. To monitor potential interactions between drug components and the packaging, the applicability of attenuated total reflectance Fourier-transform infrared (ATR-FTIR) and near-infrared (ATR-NIR) spectroscopy, differential scanning calorimetry (DSC), and X-ray diffraction (XRD) was evaluated. The mechanical strength of the packaging material after exposure to the selected formulations was assessed using a TA.XTplus texture analyzer (Stable Micro Systems, Godalming, UK).
Materials and methods
Materials
The following substances were used to prepare SEOs containing suspended drug particles: sodium cefuroxime (CEF) and vancomycin hydrochloride (VAN) (each at 5% w/w; Zhejiang Changhai Pharmaceuticals, Shaoxing, China), Miglyol®812 (fractionated coconut oil; Caesar & Loretz, Hilden, Germany), Tween® 20 (polysorbate 20, 5% w/w; Sigma-Aldrich, Steinheim, Germany), and sodium citrate (2% w/w; Sigma-Aldrich, Poznań, Poland).
Methods
Preparation of SEO formulations
The SEO carrier was prepared by mixing Miglyol® 812 with Tween® 20 at a concentration of 5% w/w. The mixture was subsequently sterile-filtered through hydrophobic polyethersulfone membrane filters (GPWP02500; Merck, Warsaw, Poland). Micronized, sterile powders of CEF and VAN (each at 5% w/w), along with sodium citrate (2% w/w), were added to the carrier, and the resulting suspension was homogenized. The following containers were included in the study: Amapack, Trzebnica, Poland (5 mL for aqueous solutions; Op1), Amapack (10 mL for oil-based solutions; Op2), Eprus, Bielsko-Biała, Poland (10 mL for aqueous solutions; Op3), Eprus (1 mL single-dose mini dropper with closure; Op4), Eprus (1 mL white bottle; Op5), and Gerresheimer, Bolesławiec, Poland (10 mL for oil-based solutions; Op6). Samples of the eye drop containers were cut into square or rectangular fragments measuring approx. from 5 × 5 mm to 10 × 10 mm. The fragments were placed in Eppendorf tubes and immersed in 0.5 mL of oil (O), oil–Tween mixture (O–T), and SEO-CEF and SEO-VAN suspensions, respectively. The prepared samples were incubated in a climatic chamber at 40°C and 60°C/75% relative humidity for 2 weeks. After incubation, the packaging material fragments were removed, gently wiped with tissue paper, and analyzed. Additionally, selected samples were washed with diethyl ether and reanalyzed after immersion in 1 mL of ether for 1 h. Structural changes and potential degradation were evaluated using FTIR and NIR spectroscopy, while DSC was employed to assess thermal properties.
Infrared and near-infrared spectroscopy (FTIR/NIR)
Infrared and near-infrared spectra were recorded using a JASCO FT/IR-4700 spectrophotometer (JASCO Co., Tokyo, Japan) equipped with a diamond attenuated total reflectance (ATR) accessory. Measurements were performed at a resolution of 4 cm–1 over the spectral ranges of 4000–400 cm–1 (FTIR) and 8200–4000 cm–1 (NIR).
Differential scanning calorimetry
The thermal behavior of the samples was evaluated using a STAR®-1 System (Mettler Toledo, Greifensee, Switzerland) equipped with an intercooler. Approximately 3–5 mg of each sample was weighed into 40 µL aluminum crucibles. Thermal analysis was performed over a temperature range from −30°C to 300°C at a heating rate of 10°C/min under a nitrogen purge at a flow rate of 60 mL/min. All measurements were normalized to sample mass.
X-ray diffraction analysis
X-ray diffraction patterns of LDPE films were recorded using an X-ray powder diffractometer (SmartLab SE; Rigaku Corporation, Tokyo, Japan). Cu Kα radiation (λ = 1.5418 Å) was used as the X-ray source. The generator was operated at 40 kV and 50 mA. Diffraction data were collected over a 2θ range of 5° to 80°, with a scanning rate of 10.00°/min and a step size of 0.01°. All measurements were performed at room temperature under constant operating conditions.
Texture analysis
The mechanical strength of container fragments was evaluated using a TA.XTplus texture analyzer (Stable Micro Systems) equipped with a 5 kg load cell. The force required to indent a spherical probe (P/5S, diameter 5 mm) into a plastic fragment (surface area approx. 1 cm2) to a depth of 300 µm was measured. Prior to testing, the probe was positioned 1.5 mm above the sample surface and then lowered at a constant speed of 0.01 mm/s. Upon contact with the sample (trigger force = 1.0 g), the probe continued to move downward to a depth of 300 µm, after which it returned to the initial position at the same constant speed. During the test, the maximum force [N] and the work of compression [mJ], defined as the area under the force–distance curve, were recorded using Texture Exponent 32 software (Stable Micro Systems).
Results
The prepared oil-based systems (SEOs) and their suspensions containing CEF and VAN were subjected to a series of analytical tests to evaluate their stability and compatibility with the selected packaging materials. Following incubation under stress conditions (40°C/75% relative humidity (RH) and 60°C/75% RH for 2 weeks), the samples were analyzed to detect changes in the active compounds and to assess potential interactions between the SEO components and the polymer matrix of the containers. A combination of complementary analytical techniques – FTIR and NIR spectroscopy, DSC, and XRD – was employed to identify structural and thermal alterations. In addition, texture analysis was performed to evaluate changes in the mechanical properties of the containers. The results provide insight into the physicochemical behavior of LDPE packaging materials following contact with oil, SEO formulations without active compounds, and SEO suspensions containing active ingredients.
Spectroscopic and thermal data
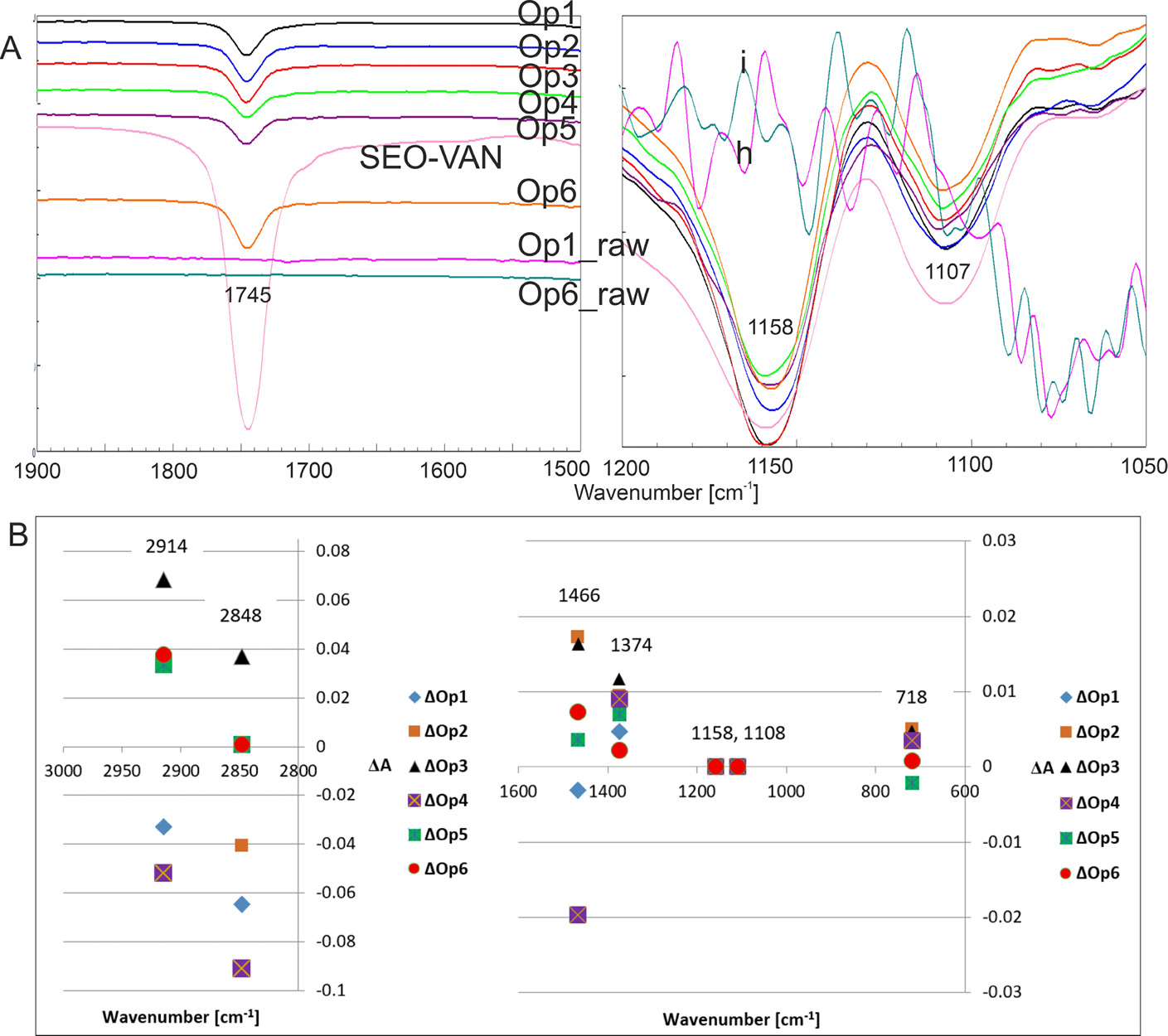
Characteristic infrared (IR) absorption bands of polyethylene (PE) (Figure 1) were observed in the mid-infrared region at 2914 cm–1 and 2848 cm–1, corresponding to asymmetric and symmetric C–H stretching vibrations of –CH2– groups, respectively. Additional bands were detected at 1469 cm–1 (–CH2 bending), 1377 cm–1 (–CH₃ bending),
1303 cm–1 (–CH2 twisting), and at 729 and 718 cm–1 (–CH₂ rocking). In the near-infrared region, bands at 5818 cm–1, 5789 cm–1, 5781 cm–1, 5771 cm–1, 5695 cm–1, 4323 cm–1, and 4252 cm–1 were attributed to overtones and combination bands of aliphatic C–H vibrations in methylene groups. The most pronounced differences between spectra of raw containers and those incubated with SEO formulations were observed at 2914 cm–1 and 2848 cm–1. Among the tested samples, only Op6 remained spectroscopically stable, showing no significant changes in these regions. The fingerprint region exhibited notable differences in absorption intensity at approx. 1745 cm–1, corresponding to ester (R–COOR) groups, which may arise from PE oxidation or from the presence of Miglyol components.7 This band was absent in the spectra of the untreated containers. After wiping and subsequent immersion in ether, 3 additional IR bands characteristic of the oil phase were identified at 1744 cm–1, 1159 cm–1, and 1109 cm–1. The presence of these bands indicates sorption of oil components onto or into the packaging material following stress exposure at 60°C/75% RH for 2 weeks. According to the European Pharmacopoeia,9 PE exhibits 2 characteristic bands at 1471 cm–1 and 1465 cm–1. However, all tested containers displayed a single band at 1466 cm–1, consistent with LDPE.10
Differential scanning calorimetry
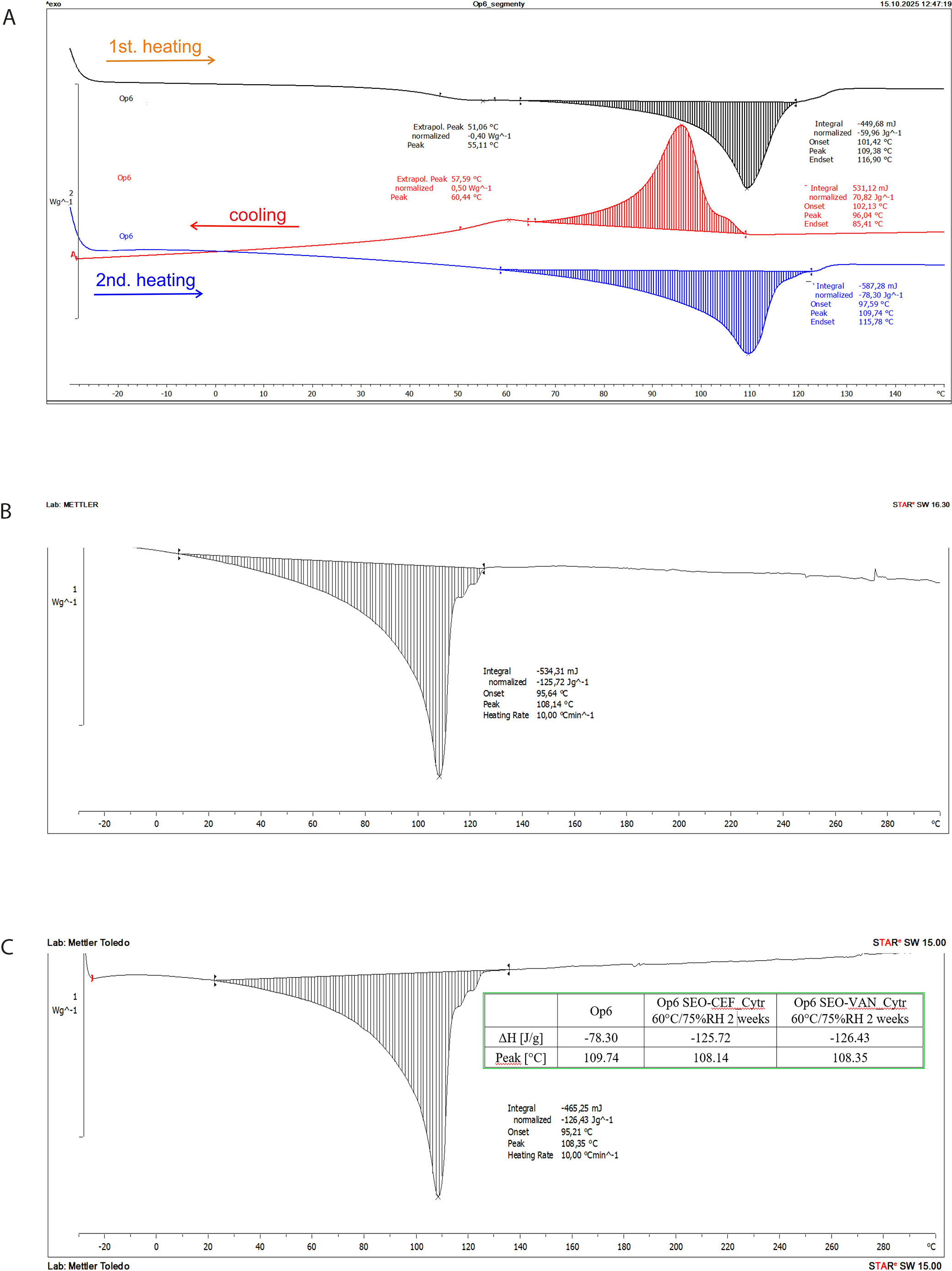
The melting endotherms of LDPE containers (Op1–Op6) were observed in the temperature range of 108.69–116.51°C (Table 1). During the 2nd heating cycle, the small endothermic peaks detected in the 1st heating run within the 40–50°C range were no longer present (Figure 2). Differences in the α-relaxation temperatures were observed among the individual container types and following incubation of the packaging materials in SEO at ambient temperature. Variations in these peaks between containers suggest differences in their thermal history and structural organization. No statistically significant differences in melting enthalpy (ΔHm) were observed between the tested samples (Table 1), indicating comparable degrees of crystallinity. Low-density polyethylene is a semi-crystalline polymer composed of crystalline domains dispersed within amorphous regions. In the 1st heating run, a small endothermic peak observed between 40°C and 60°C can be attributed to relaxation processes in the amorphous phase. These phenomena are consistent with the viscoelastic behavior of PE and correlate with internal friction peaks reported in the literature.11, 12, 13, 14 Plasticization of the amorphous regions may reduce stiffness, increase deformability, and alter mechanical strength, findings that are supported by the texture analysis results. After storage in SEO formulations at elevated temperatures, only negligible changes in the thermal characteristics of the containers were observed. During the 1st heating cycle, the peak temperatures were generally slightly lower than those of the untreated (raw) containers. However, in the case of Op4 incubated with SEO, an increase in peak temperature of approx. 10% was noted (Table 1). No significant differences were detected during the 2nd heating cycle. Overall, after storage under stress conditions (60°C/75% RH for 2 weeks) in SEO formulations containing APIs and sodium citrate, no evidence of substantial thermal interactions or structural alterations in the packaging materials was observed.
Mechanical integrity and drug compatibility
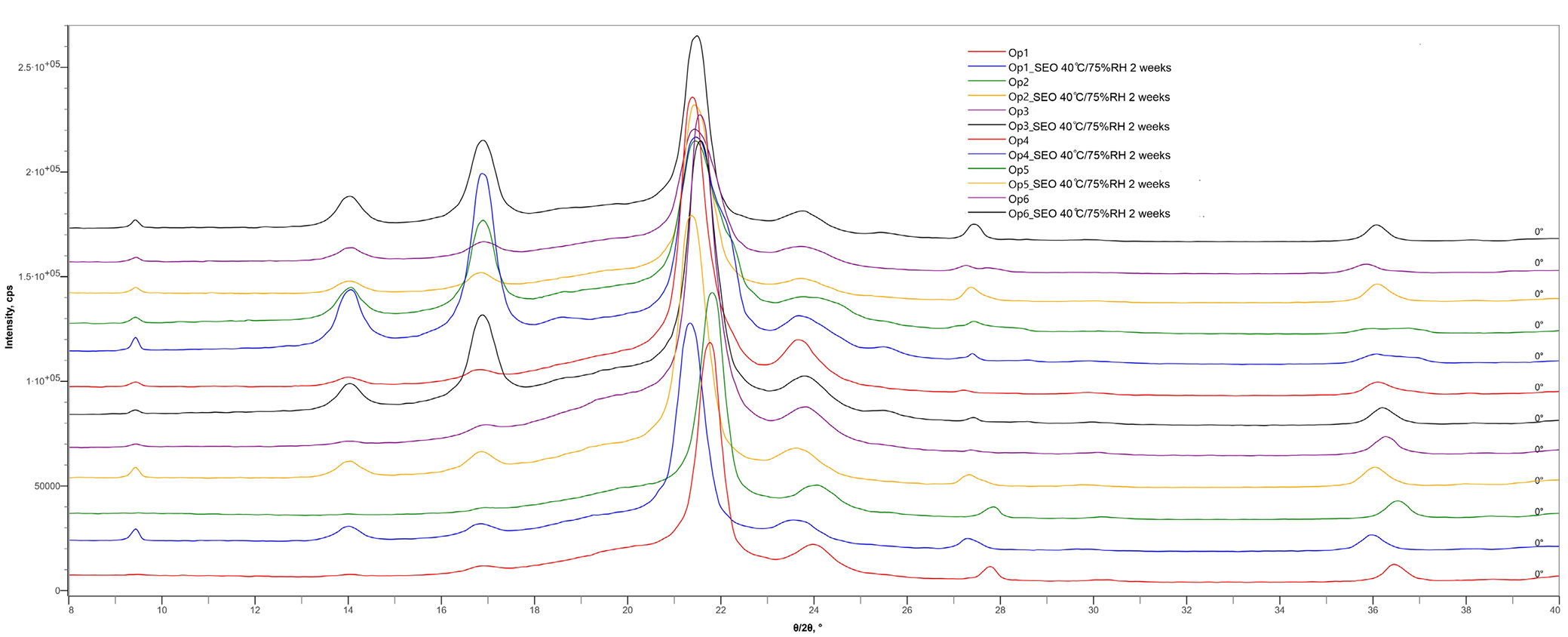
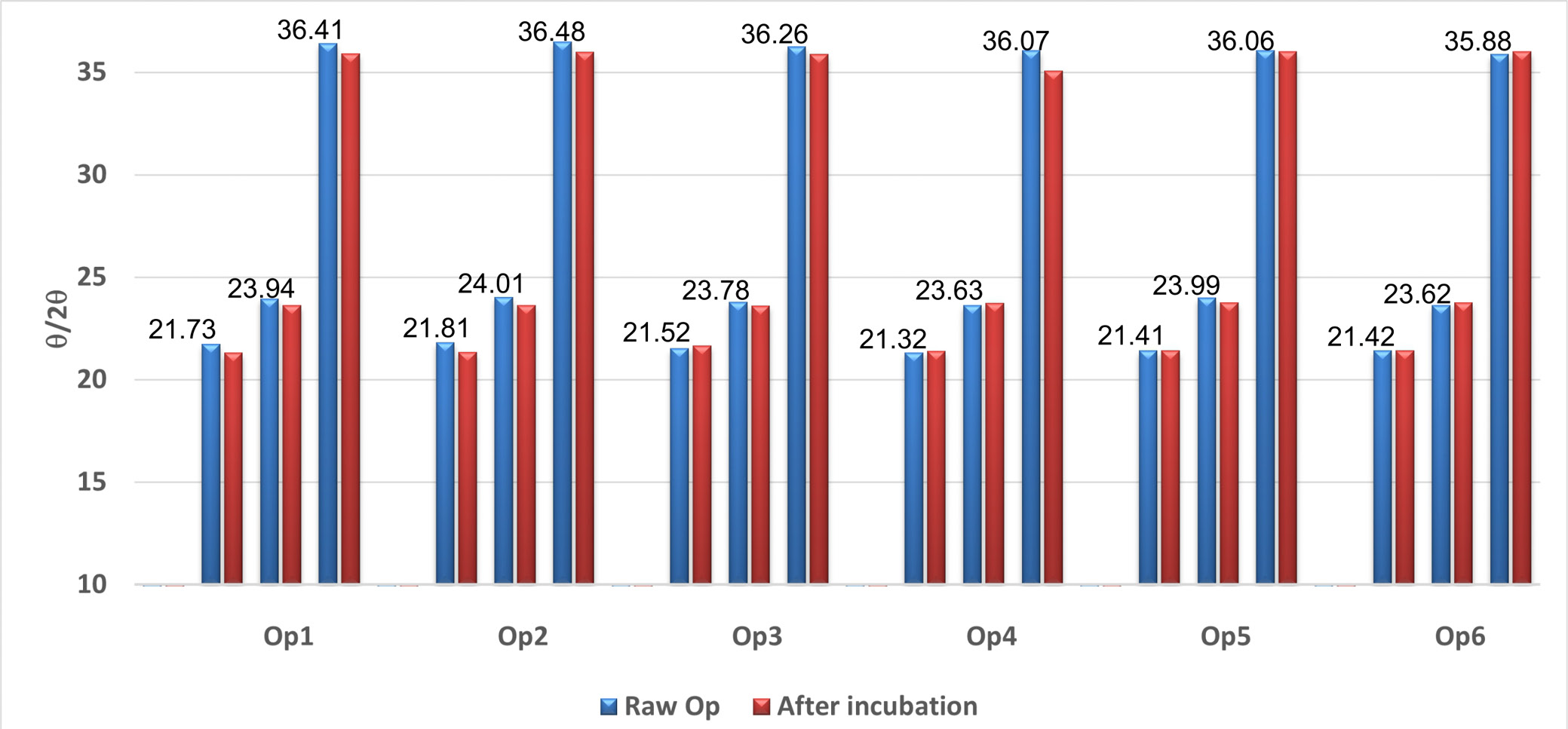
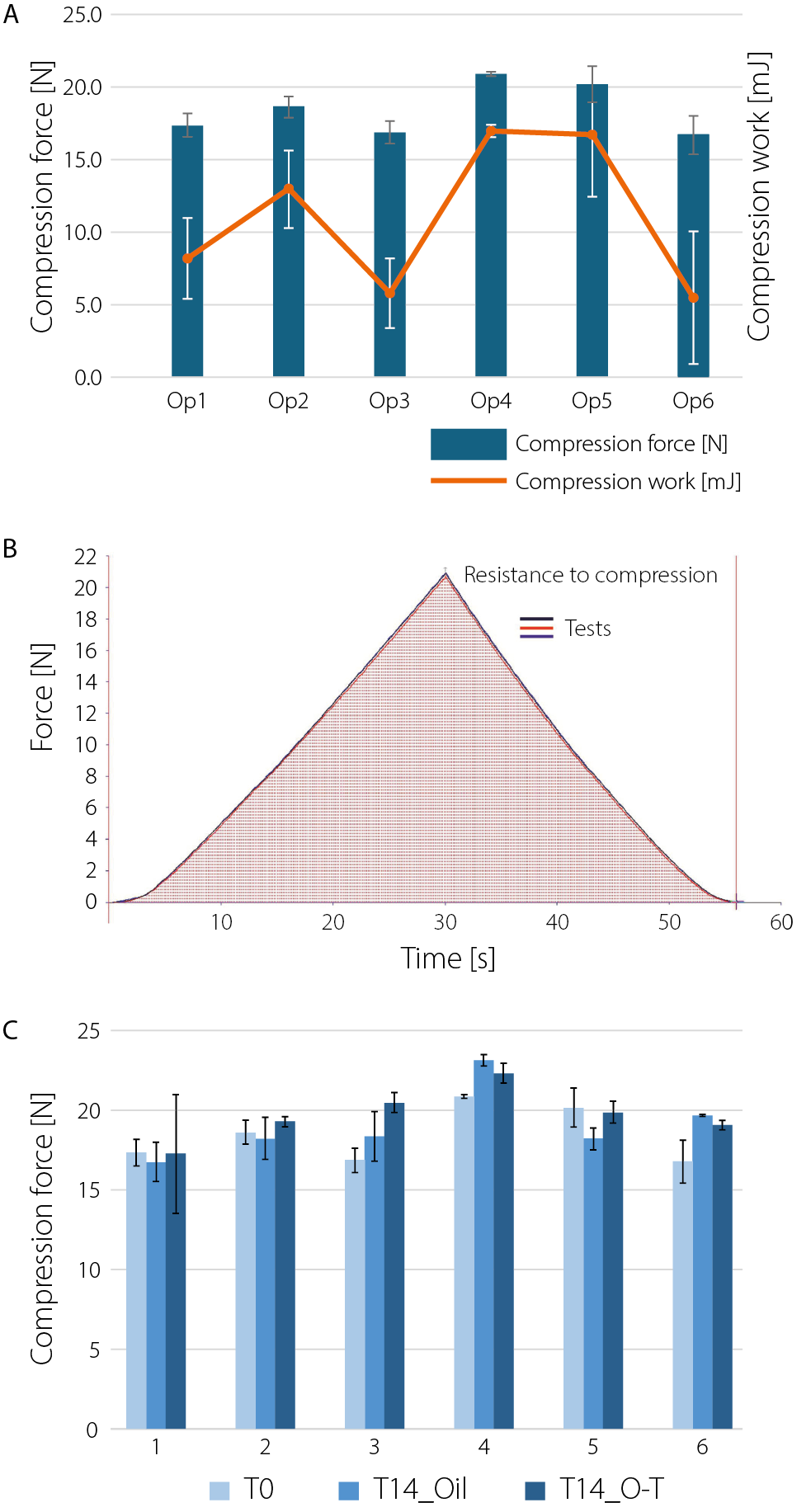
The XRD patterns of LDPE are shown in Figure 3. Peaks between 13° and 19° (2θ) are visible and correspond to reflections also recorded for the tape used to hold the container fragments in the instrument. The 2θ angles for LDPE were observed at 21.3°, 28.43°, and 36.04°. After incubation of the packaging with SEO, the XRD method indicated subtle θ/2θ shifts toward smaller angles, which may indicate an increase in the distance between crystalline planes (d-spacing). The plasticizer or oil can penetrate between the chains, pushing the structure apart, which was demonstrated during tests using the texturometer and FTIR. The differences in 2θ angles are mainly due to changes in the degree of crystallinity of LDPE after interaction with SEO (oil). The values of the shifts between the angles of the raw polymer and those of Op after incubation are presented in Figure 4. For polymeric packaging composites, diffraction peaks were observed at 2θ = 21.3° and 23.8°, corresponding to the (110) and (200) crystallographic planes, respectively, which has also been confirmed in the literature.15 The graph below (Figure 5) presents the results of the mechanical compression strength test of the tested packaging fragments, expressed as the maximum compression force [N]. Raw containers are not identical (Figure 5), but the data obtained during testing of the mechanical properties of selected package fragments (compression force at time T0) are similar (Figure 5). Regardless of the bottle type, their average values ranged from 17 N to 21 N (compression force) and from 3.7 mJ to 5.1 mJ (compression work). We observed that the ease of penetration of the spherical probe into the package fragments depends on their thickness. The highest compression force and work were measured for bottles with the thinnest walls (500–600 µm) – Op4 (Figure 5, Figure ). Although the tested package fragments were relatively small (approx. 1 cm2), the wall thickness in different locations within a given fragment varied by up to approx. 100 µm (approx. 15%). For this reason, the results of mechanical measurements within a single fragment show considerable variability: the relative standard deviation (RSD) for compression work was 13–15% (Op1–Op3 stored in oil for 14 days, T14_Oil), and even 21.5–36.6% (force and compression work for Op1 stored for 14 days in the O–T mixture, T14_O–T).
Discussion
The stability and compatibility of ophthalmic formulations with their primary packaging are critical factors in ensuring the safety and efficacy of drug delivery systems. This study evaluated the interactions between 3 distinct systems – pure oil, an oil-based self-emulsifying carrier (SEO), and SEO containing suspended CEF and VAN – and commercially used eye drop containers.
Interpretation of spectroscopic and thermal data
These findings align with observations by Garrido-López et al.,16 who described the migration of lipophilic components into PE. While such “scalping” can often lead to the loss of active ingredients or excipients, our data suggest a potential secondary benefit: the diffusion of oils into PE may act as a plasticizer, potentially enhancing chemical and oxidative stability or imparting radiopacity for specialized medical applications. Structural and thermal changes occurring in the samples after exposure to elevated temperature and humidity, simulating accelerated aging conditions, were analyzed using FTIR, NIR, DSC, XRD, and texture analysis to evaluate potential interactions between the formulation components and the packaging materials. The results obtained in this study enable the assessment of the influence of SEO composition and packaging type on the stability and safety of ophthalmic formulations. Fourier-transform infrared spectroscopy provided crucial evidence regarding the surface interactions between the lipophilic SEO and the polymer matrix. The identification of characteristic bands at 1744 cm–1, 1159 cm–1, and 1109 cm–1 after stress testing confirms the formation of a persistent Miglyol-based film on the packaging surface. The fact that this residue remained after chemical cleaning (ether soaking) suggests a high affinity between the oil and the polyolefin chains. Differential scanning calorimetry analysis further supported these observations. The consistency of melting profiles between raw and stressed containers indicates that the bulk thermal properties of the packaging remain intact. However, the subtle shifts of melting peaks observed in LDPE may suggest changes in the packing density of the amorphous phase, indicating that while the crystalline structure remains stable – as confirmed by the lack of significant changes in XRD diffraction peak intensities – the amorphous regions are susceptible to subtle structural rearrangements due to oil and polysorbate 20 penetration after 2 weeks at 60°C/75% RH.
The migration and adsorption of lipophilic cosmetic ingredients into polyolefins have already been confirmed using FTIR and DSC methods by Bolte et al.17 for PE packaging. In this context, sorption (“scalping”) phenomena, i.e., the adsorption or absorption of substances onto the surface or within the polymer matrix, may occur. In all samples tested, no noticeable changes in the diffraction peak intensities or in the widths of the crystalline peaks were observed. However, differences in peak positions during the first heating, even if not significant, may indicate variations in the crystallinity of the composite materials. Structural modifications in polymers – such as altered chain mobility – can occur after the introduction of oils, which is also manifested by subtle shifts in the α-relaxation temperatures in DSC curves.18 Researchers attribute these processes to changes in the packing density of the amorphous phase.18
Mechanical integrity and drug compatibility
Texture analysis confirmed that the structural integrity of the containers was not compromised. Storing the packaging in SEO at elevated temperature (40°C) for 14 days did not significantly alter the mechanical strength parameters. The variations observed in probe penetration were primarily a function of wall thickness rather than material degradation induced by the SEO carrier. This is a vital finding for the pharmaceutical industry, as it ensures that the physical protection provided by the bottle remains consistent throughout the product’s shelf life.
Clinical implications of active ingredient stability
The most significant finding of this study is the lack of interaction between the suspended APIs – CEF and VAN – and the packaging materials. More advanced methods, such as inductively coupled plasma–mass spectrometry (ICP–MS) or liquid chromatography–mass spectrometry (LC–MS), may be required to confirm the absence of APIs on the surface of the container.9 The absence of drug detection within the polymer matrix suggests that the SEO system effectively sequestered these hydrophilic salts, preventing their migration or adsorption. This ensures that the concentration of the antibiotics remains stable within the formulation, which is paramount for the treatment of severe ocular infections where precise dosing is required.
Limitations
This study has several limitations that should be
acknowledged. One of the main constraints of the XRD analysis arises from the geometry of the packaging samples. The shape and curvature of the packaging materials may have affected the diffraction conditions, potentially influencing peak intensity and signal quality. As a result, the obtained data should be interpreted with caution.
Furthermore, to obtain a more comprehensive understanding of the interactions between the packaging materials and the oil-based suspension matrix, further investigations are warranted. In particular, Raman confocal microscopy mapping could provide spatially resolved information on chemical changes and diffusion phenomena, while ICP-MS analysis would allow for sensitive detection of potential elemental migration. These complementary techniques would contribute to a more complete characterization of packaging–matrix interactions.
Conclusions
Physical and chemical characterization of LDPE containers was carried out to determine changes in their structural, thermal, functional group, and crystalline or amorphous properties after storage and incubation in O, O–T (SEO), SEO-CEF, and SEO-VAN under stress storage conditions. Basic characterization of PE containers enables the recording and understanding of their behavior in contact with the formulations. Fourier-transform infrared spectra allow observation of negligible interactions, visible as small (up to 8 cm–1) band shifts. In general, independent of the active compound tested (CEF and VAN), only interactions related to the presence of O (oil) and contact with the containers under stress conditions were observed. Using the DSC method, small but detectable differences in the α-relaxation temperatures and melting enthalpy of LDPE containers were observed after stress testing. This research aimed to investigate the fundamental interactions between LDPE and SEO in the context of degradation studies. Oil–packaging and oil–surfactant interactions were minimal and consistent. No oil–surfactant incompatibilities were observed. The findings of this study may be incorporated into the registration documentation, specifically within the “Quality” section (Module 3) of the CTD dossier. The polymeric materials that demonstrated stability under the tested conditions may be selected for the storage of commercial innovative SEO products.
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Consent for publication
Not applicable.
Use of AI and AI-assisted technologies
Not applicable.